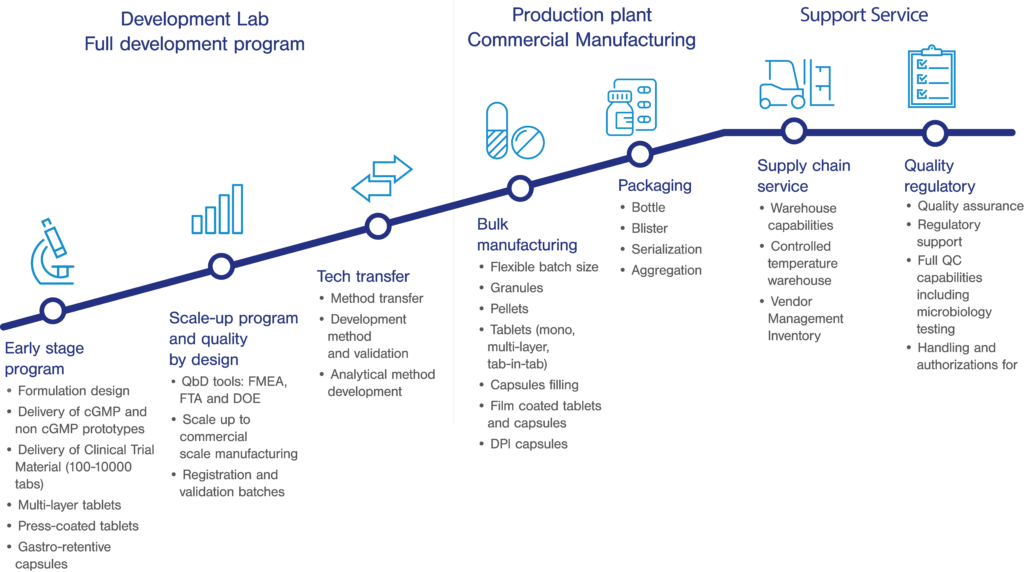
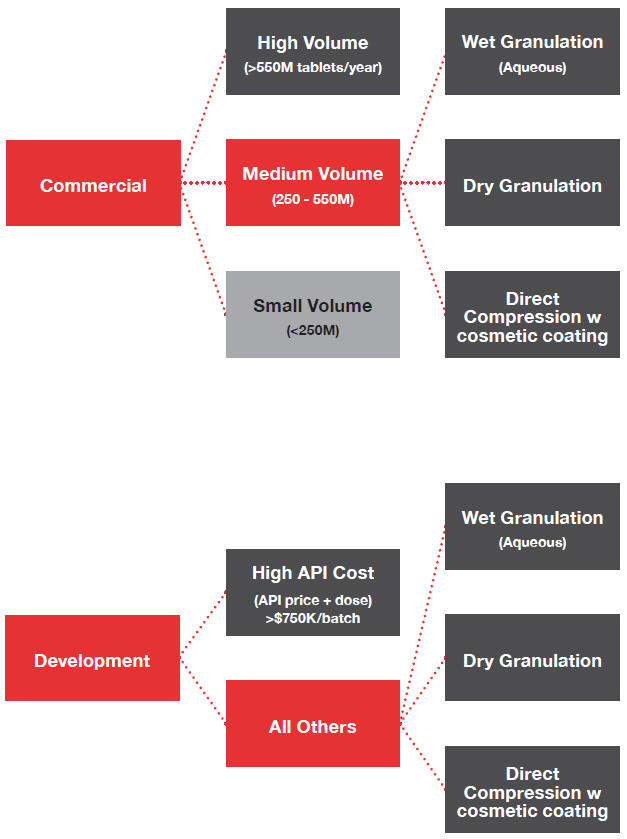
Clinical Trial Supply Management | Discover How Outsourcing Clinical Batch Manufacturing Can Prevent Supply Issues | UPM Pharmaceuticals - UPM Pharmaceuticals
![PDF] PLANNING PROCESS OF PILOT BATCH PRODUCTION OF AN INNOVATIVE DRUG FOR CLINICAL TRIAL IN A PHARMACEUTICAL INDUSTRY | Semantic Scholar PDF] PLANNING PROCESS OF PILOT BATCH PRODUCTION OF AN INNOVATIVE DRUG FOR CLINICAL TRIAL IN A PHARMACEUTICAL INDUSTRY | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/68331731d65affa1bc7302b1885c4746cd853691/6-Figure3-1.png)
PDF] PLANNING PROCESS OF PILOT BATCH PRODUCTION OF AN INNOVATIVE DRUG FOR CLINICAL TRIAL IN A PHARMACEUTICAL INDUSTRY | Semantic Scholar
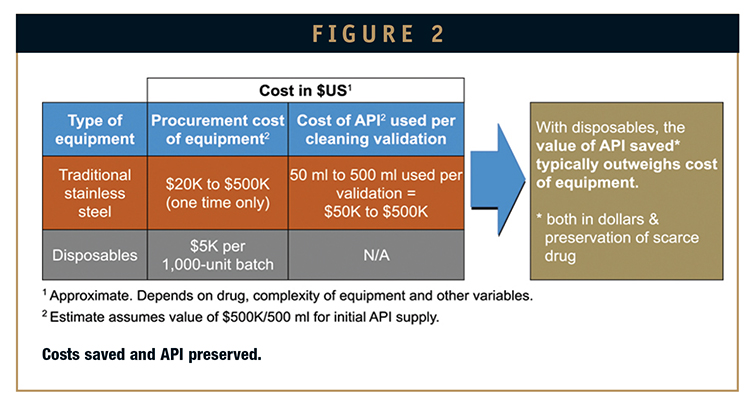
DISPOSABLE TECHNOLOGY - Use of Disposable Technology in Clinical Fill & Finish Manufacturing: Benefits & Considerations

Impact of COVID-19 on Manufacturing of Cell and Gene Therapy and Biotech Products, and Overall Clinical Trial Landscape | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Main steps of drug production process for clinical study. Source: The... | Download Scientific Diagram

Frontiers | From Single Batch to Mass Production–Automated Platform Design Concept for a Phase II Clinical Trial Tissue Engineered Cartilage Product | Medicine

Considerations for Manufacturing Investigational Medicinal Products for Clinical Studies - Clinical Trials Arena
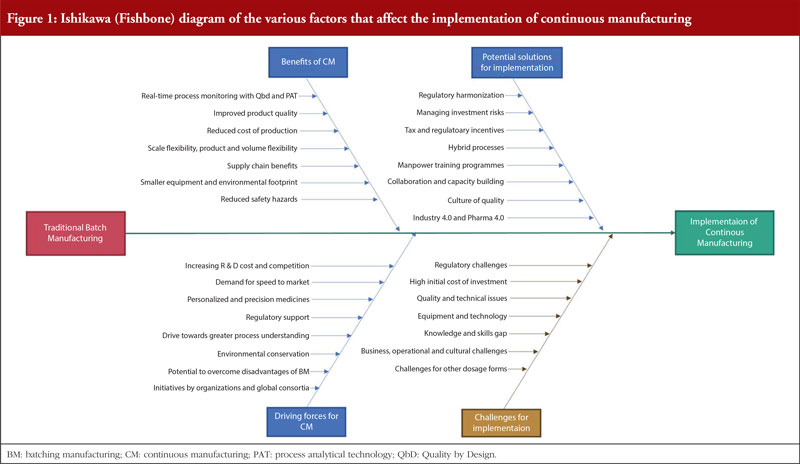
Continuous manufacturing versus batch manufacturing: benefits, opportunities and challenges for manufacturers and regulators - GaBI Journal
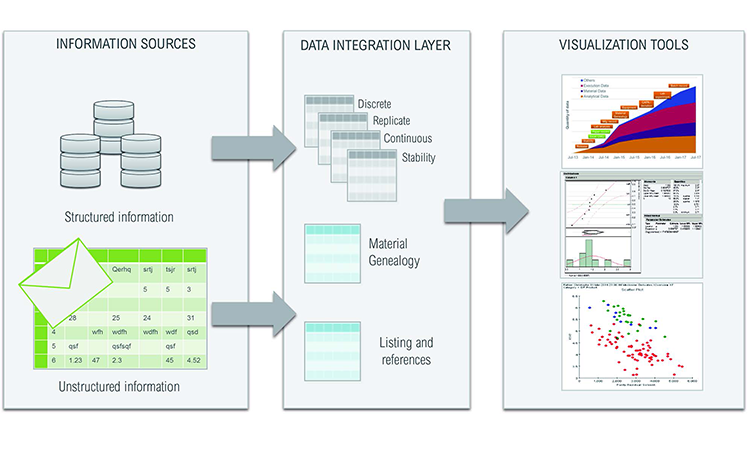
Finding Relationships Between Clinical Batch Quality Data & Patient Outcomes | Pharmaceutical Engineering

Clinical Trial Supply Management | Discover How Outsourcing Clinical Batch Manufacturing Can Prevent Supply Issues | UPM Pharmaceuticals - UPM Pharmaceuticals
How Supply Chain Digitalization Efforts Will Help Increase the Efficiency of Drug Approvals - ACD/Labs Blog
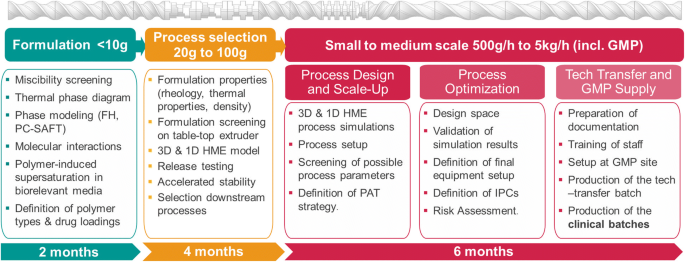
Developing HME-Based Drug Products Using Emerging Science: a Fast-Track Roadmap from Concept to Clinical Batch | SpringerLink
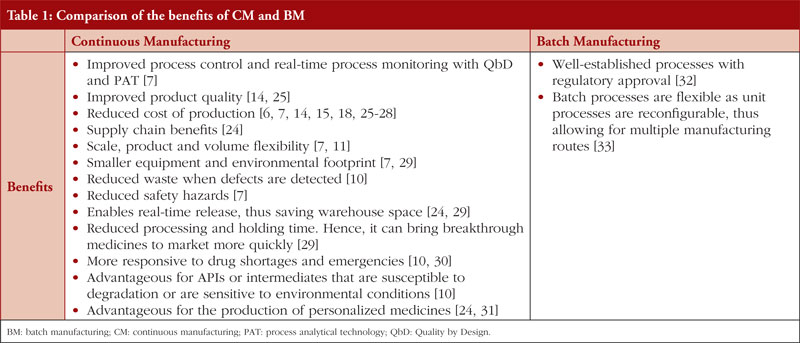
Continuous manufacturing versus batch manufacturing: benefits, opportunities and challenges for manufacturers and regulators - GaBI Journal