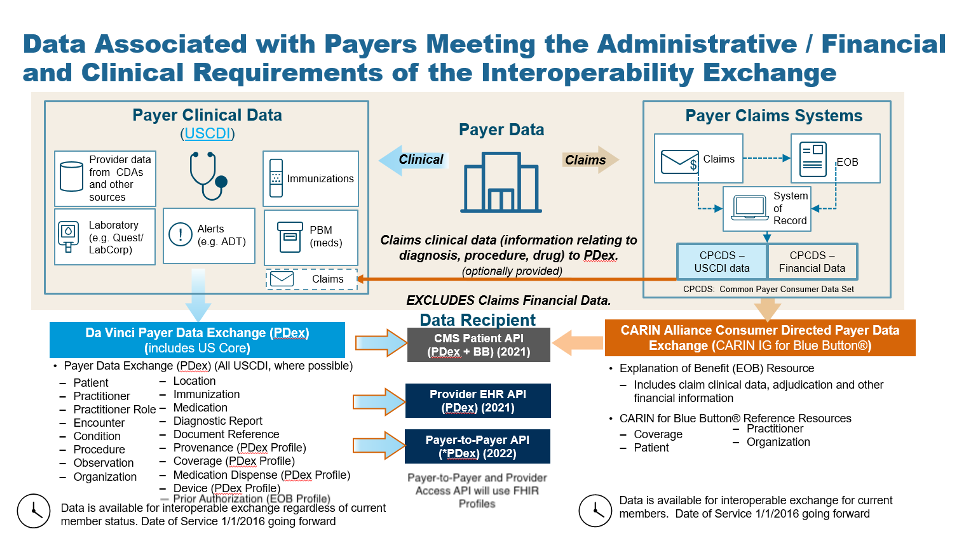
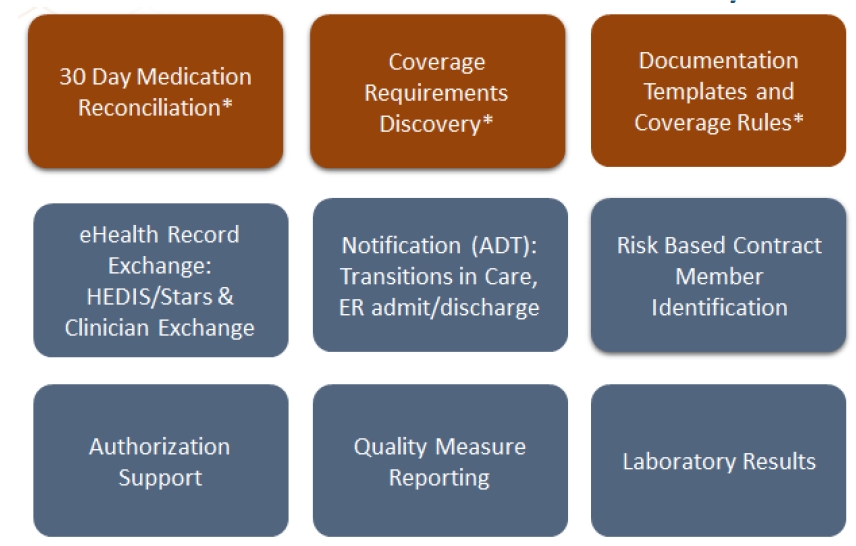
Centauri Health Solutions Adds Clinical Data Exchange Capabilities with Acquisition of Secure Exchange Solutions

Secure Exchange Solutions Automates Clinical Data Analysis Evaluation to Help Solve the Prior Authorization Challenge for Provider and Payer CIOs - Secure Exchange Solutions

Standardizing data exchange for clinical research protocols and case report forms: An assessment of the suitability of the Clinical Data Interchange Standards Consortium (CDISC) Operational Data Model (ODM) | Semantic Scholar

Benefits of Clinical Data Interchange Standards Consortium (CDISC) to Healthcare IT | Covetus Technologies Pvt Ltd