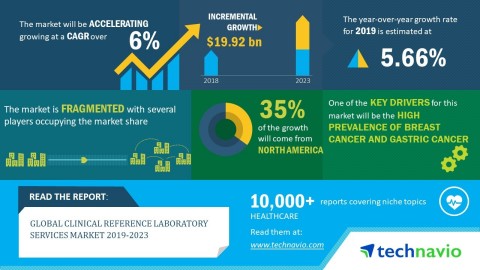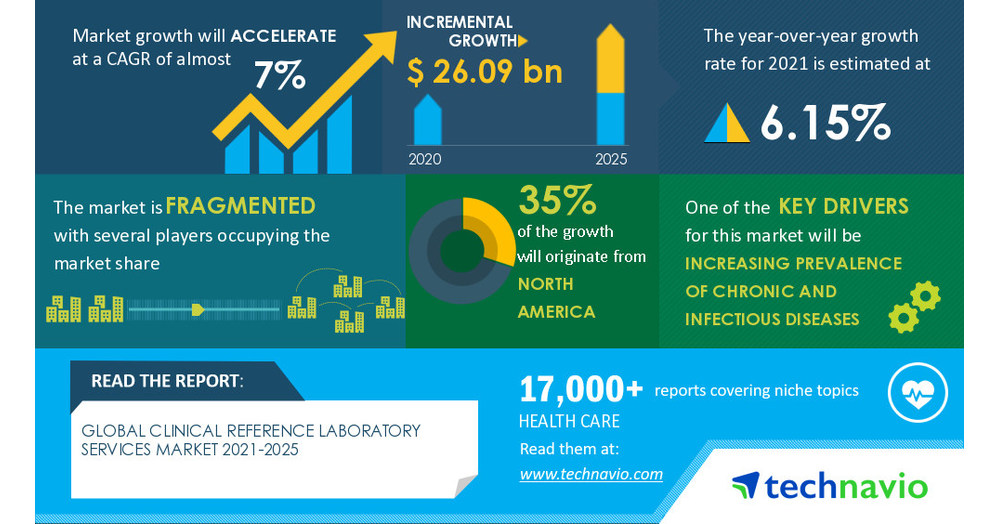
Clinical Reference Laboratory Services Market to garner USD 26.09 Billion with almost 7% CAGR during 2021-2025: Technavio

Benefits of Liquid Stable Clinical Reference Materials for Clinical Laboratories | Clinical Lab Manager

Clinical Reference Laboratory Makes First At-Home COVID-19 Saliva Test Available Through Walgreens Find Care®

Clinical Reference Laboratory Receives FDA Emergency Use Authorization for Best-in-Class Self-Collected COVID-19 Saliva Test

Clinical Reference Laboratory buys testing company Confirm BioSciences - Kansas City Business Journal

New CRL COVID-19 Research: Viral Load in Asymptomatic vs. Symptomatic Cases is Similar - Clinical Reference Laboratory

Clinical Reference Laboratory to Market Most Cost-Effective, Gold-Standard Self-Collected COVID-19 Saliva Test Directly to Consumers

Methodological Considerations for Implementation of Lymphocyte Subset Analysis in a Clinical Reference Laboratory - MUIRHEAD - 1986 - Annals of the New York Academy of Sciences - Wiley Online Library