Book M1: 2022 Mini Pocket-Sized (3" x 5") ICH Guidelines for GCP (E6) – Clinical Research Resources, LLC

Drug Safety Archives - Qtech-Sol USA offers Clinical Research / Trials, Pharmacovigilance, Drug Safety, Clinical Data Management, Clinical SAS Programming and Healthcare BA Training Programs

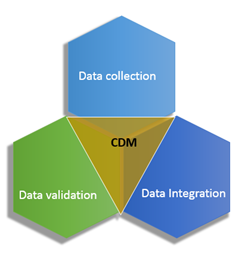
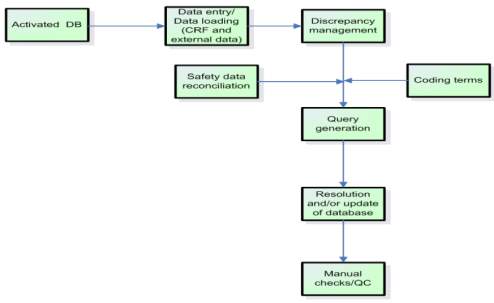
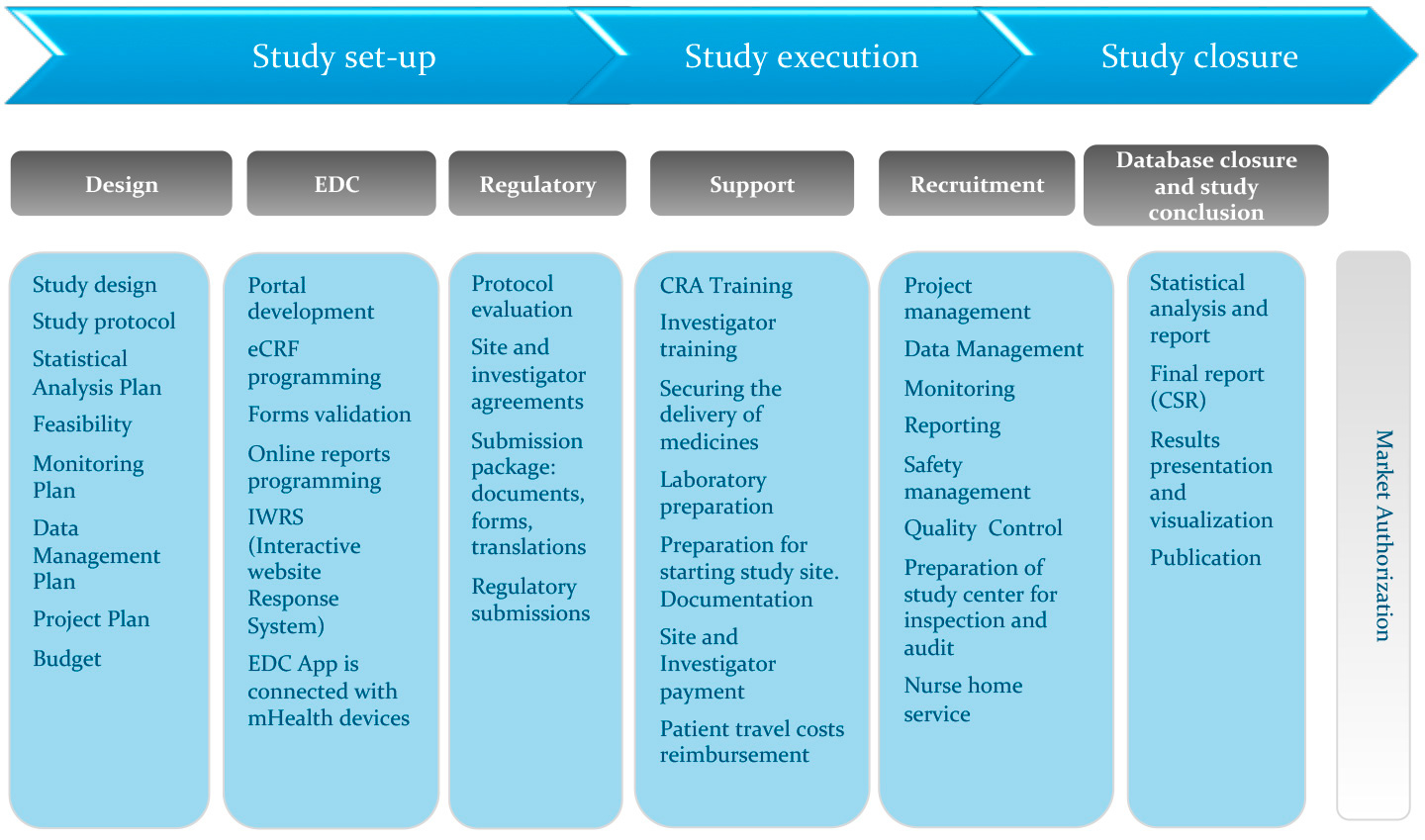
PDF) Clinical data management: Current status, challenges, and future directions from industry perspectives

Clinical Safety Data Management: Definitions and Standards for Expedited Reporting (ICH E2A) — Clinical Pathways

Clinical Safety Data Management: Definitions and Standards for Expedited Reporting (ICH E2A) — Clinical Pathways

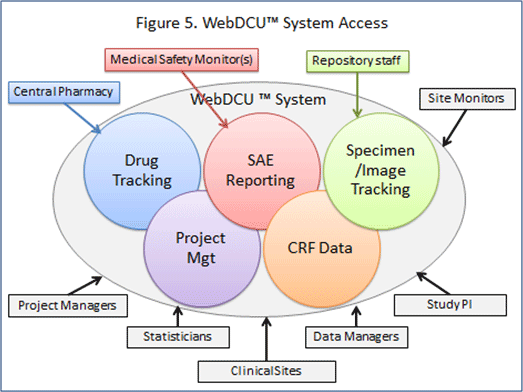
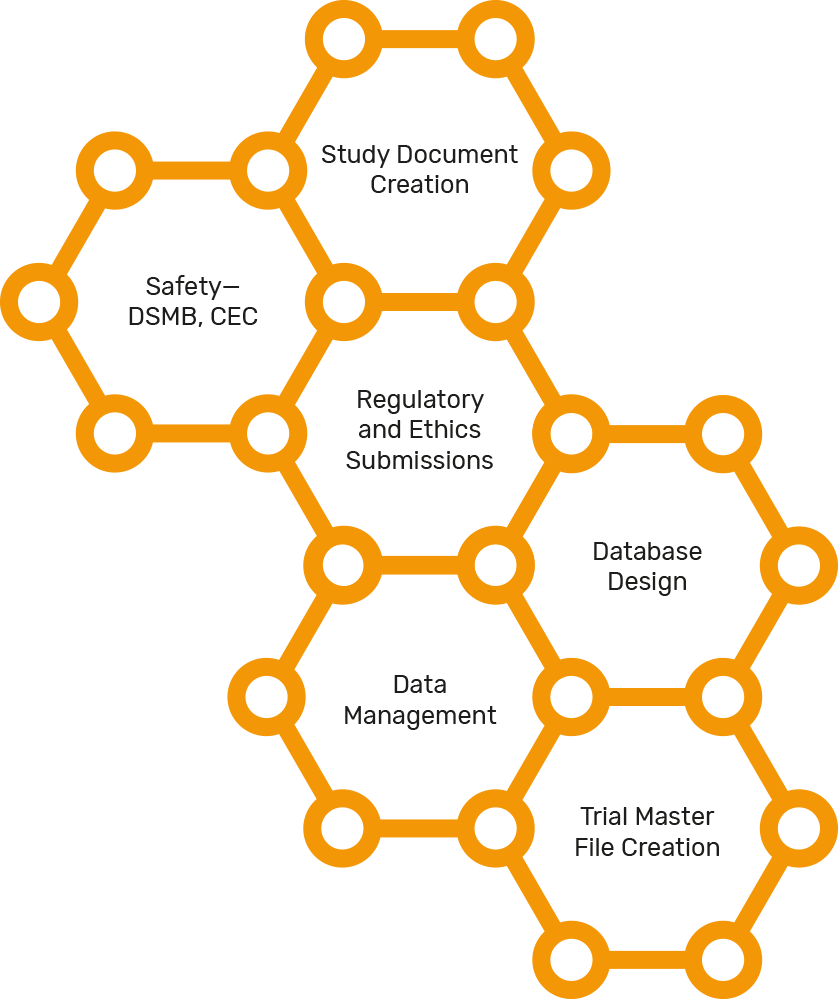
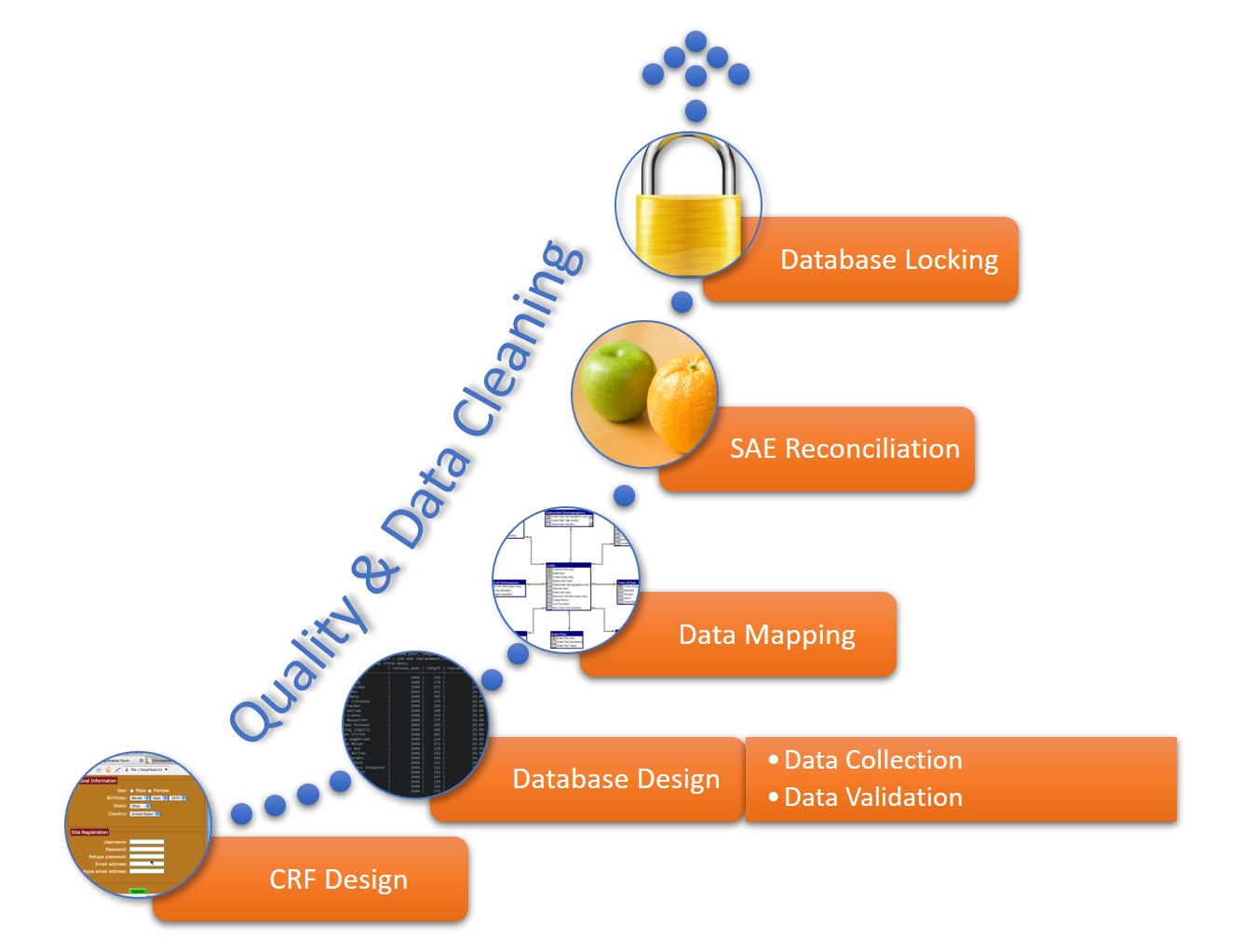
Clinical Safety Data Management | Pharmacovigilance Consulting Services | Quality Data Services, Inc

Clinical Safety Data Management | Pharmacovigilance Consulting Services | Quality Data Services, Inc