Multiple Co-primary Endpoints: Medical and Statistical Solutions A Report From the Multiple Endpoints Expert Team of the Pharmac
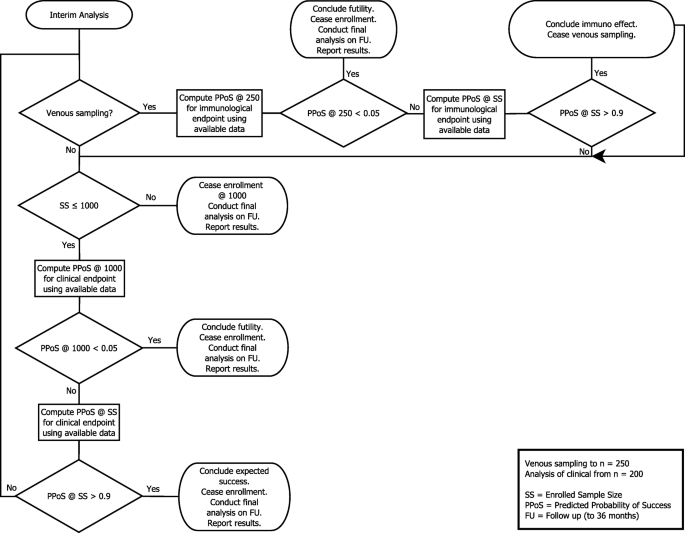
The ORVAC trial: a phase IV, double-blind, randomised, placebo-controlled clinical trial of a third scheduled dose of Rotarix rotavirus vaccine in Australian Indigenous infants to improve protection against gastroenteritis: a statistical analysis
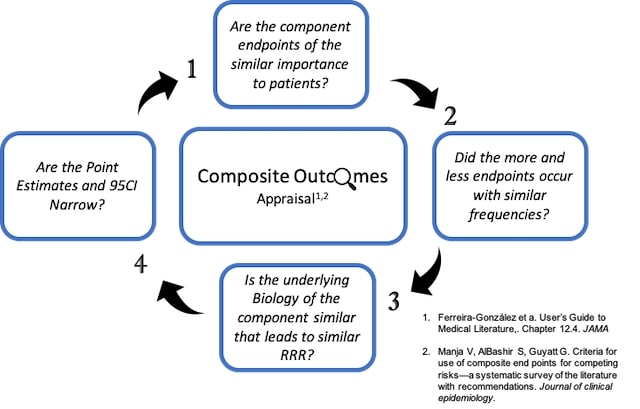
A study design with conditional, serially assessed coв•'primary endpoints: An application to a singleв•'arm, pilot non
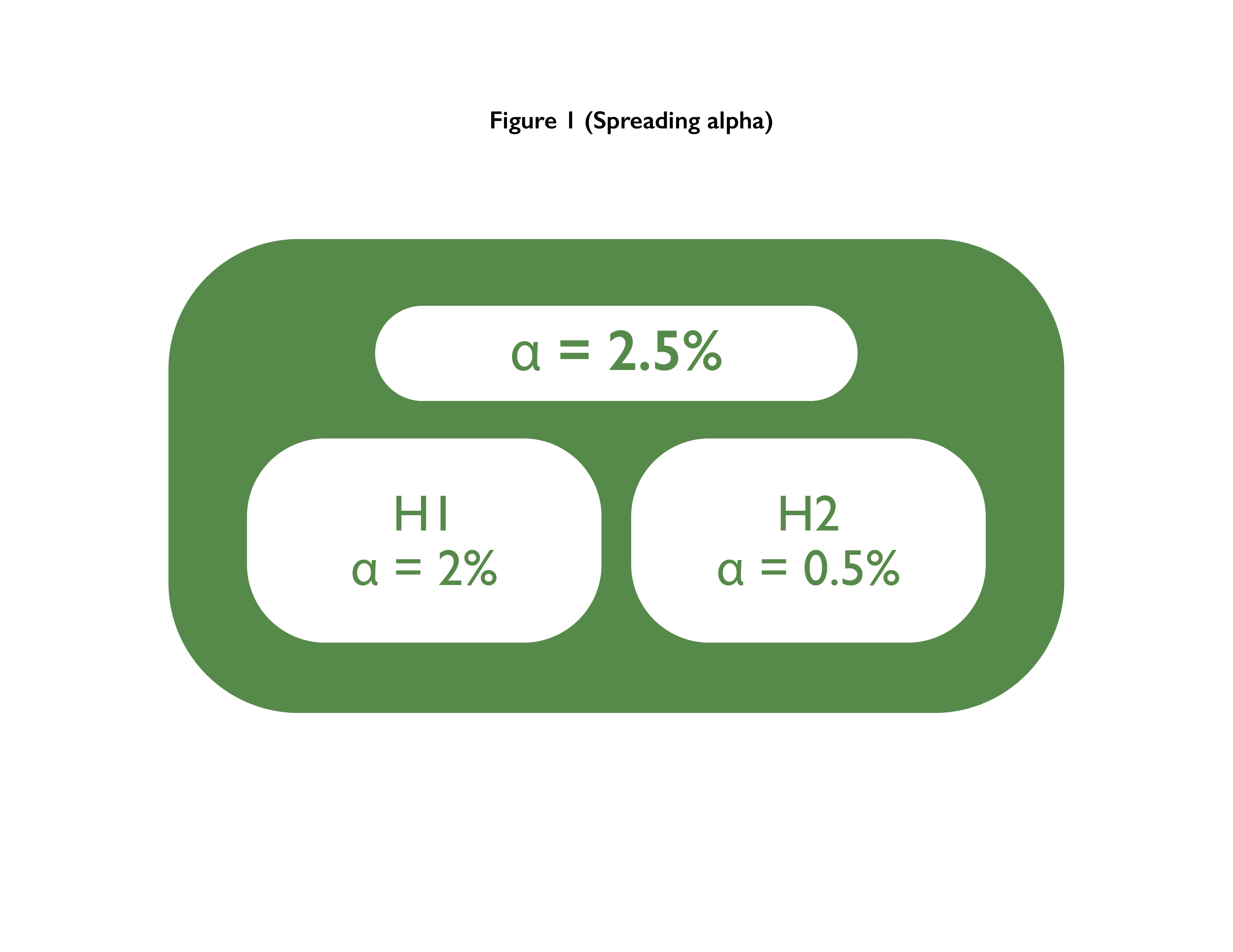
Multiple Co-primary Endpoints: Medical and Statistical Solutions A Report From the Multiple Endpoints Expert Team of the Pharmac

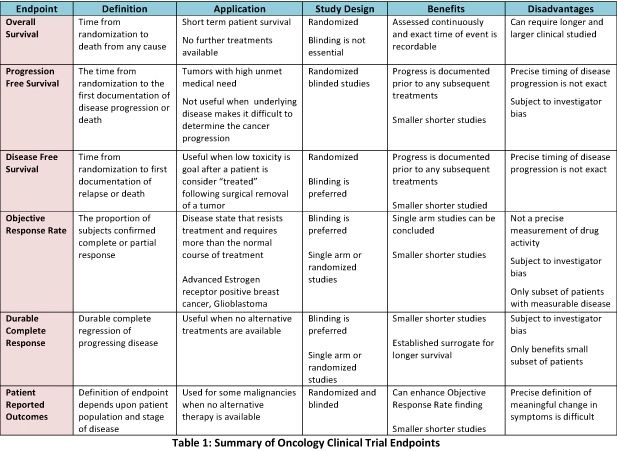
Choice of Primary (or Co-primary) Endpoints: Efficacy, Safety, or Net Clinical Benefit in Superiority and Non-inferiority Trials | tctmd.com
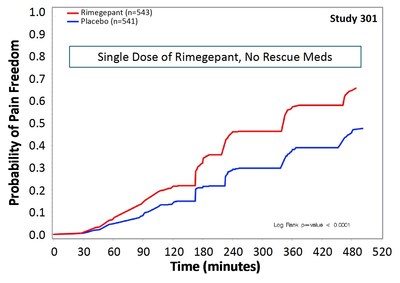
Biohaven Announces Successful Achievement of Both Co-Primary Regulatory Endpoints in Two Pivotal Phase 3 Trials of Rimegepant an Oral CGRP Receptor Antagonist for the Acute Treatment of Migraine | Biohaven Pharmaceuticals
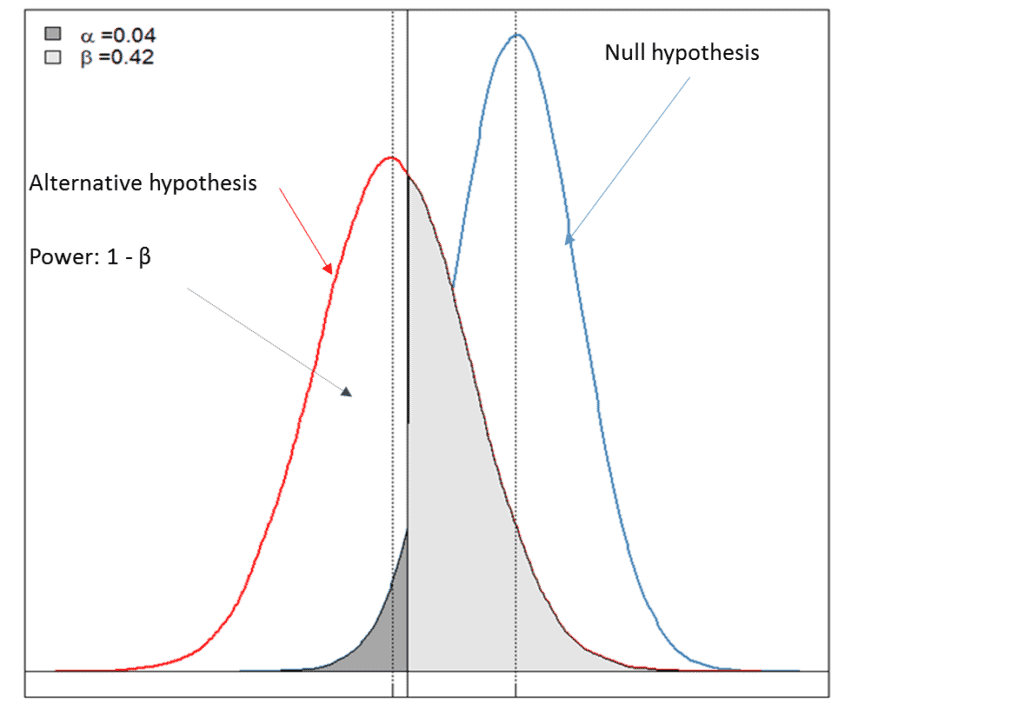
Design, data monitoring, and analysis of clinical trials with co-primary endpoints: A review: Journal of Biopharmaceutical Statistics: Vol 28, No 1