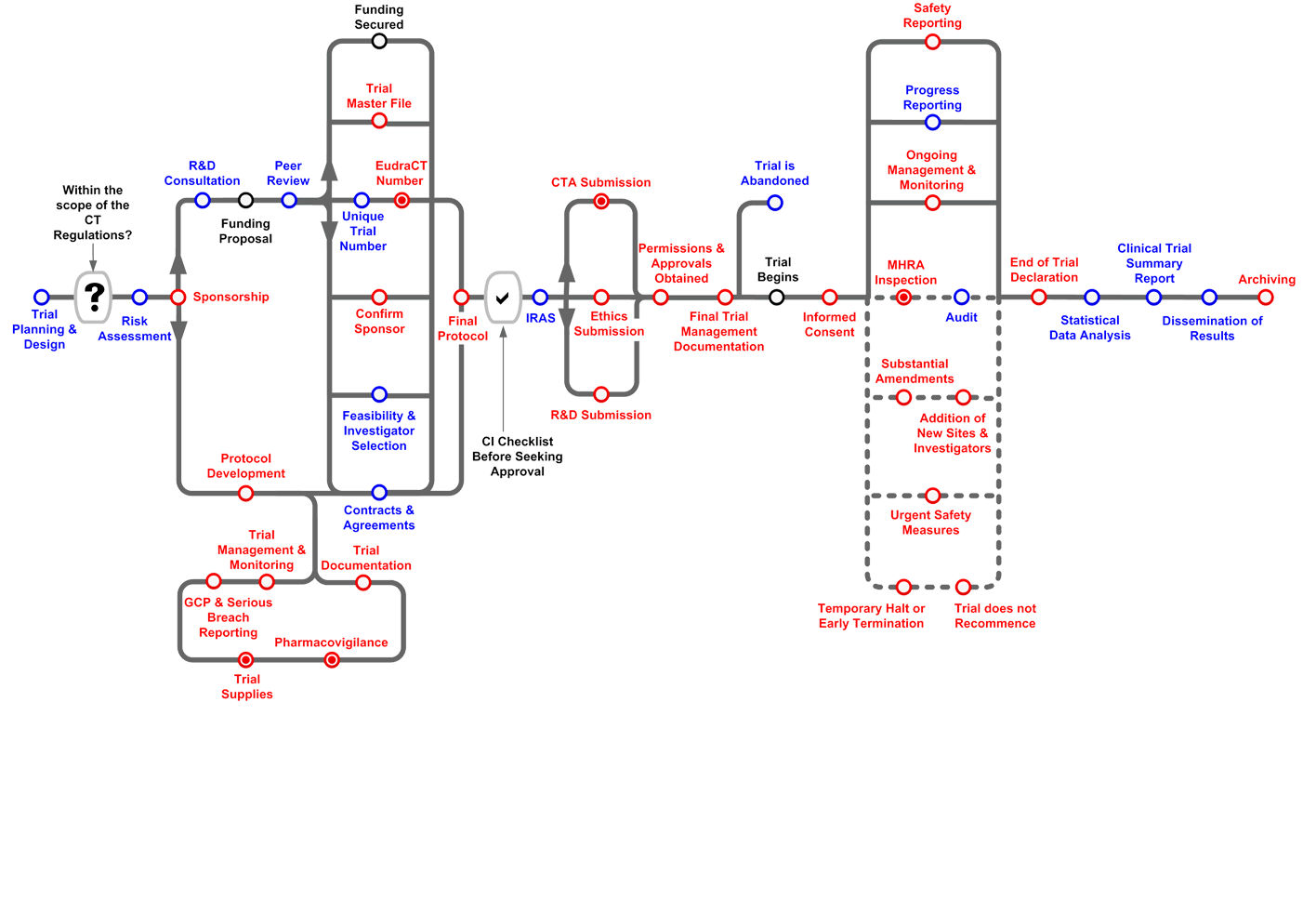
Book 6: 2021 Clinical Trials in The EU: Selected Legislation, Guidelin – Clinical Research Resources, LLC

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - The Lancet Digital Health
1/15 KLH-22 version 4 Requirements Governing the Text of Patient Information Leaflet Trial Subject Information Sheet /Informed C
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health
![PDF] Clinical trial information: developing an effective model of dissemination and a framework to improve transparency | Semantic Scholar PDF] Clinical trial information: developing an effective model of dissemination and a framework to improve transparency | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0a20197396633037aa0f5903030bcb5edb22137/152-Table20-1.png)
PDF] Clinical trial information: developing an effective model of dissemination and a framework to improve transparency | Semantic Scholar
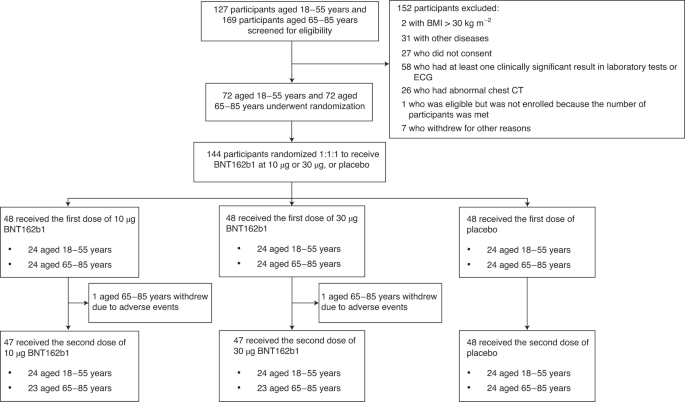
Safety and immunogenicity of the SARS-CoV-2 BNT162b1 mRNA vaccine in younger and older Chinese adults: a randomized, placebo-controlled, double-blind phase 1 study | Nature Medicine
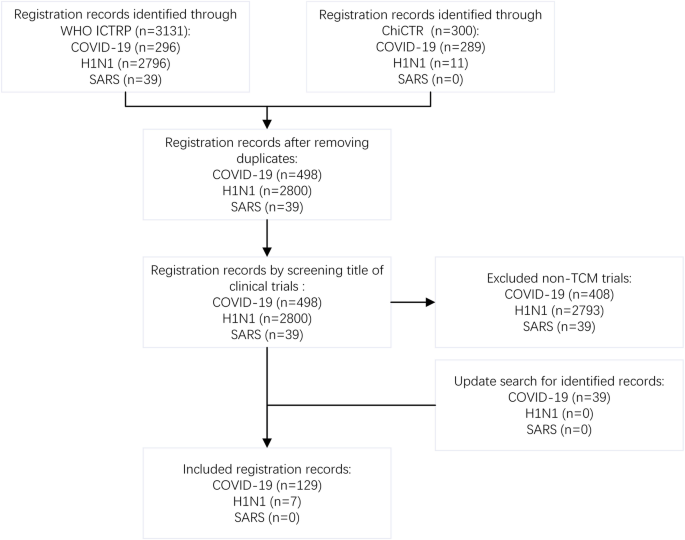
Calling for improved quality in the registration of traditional Chinese medicine during the public health emergency: a survey of trial registries for COVID-19, H1N1, and SARS | Trials | Full Text

PDF) Qualitative features in clinical trials: coordinates for prevention of passive and active misconduct