Acceptance of surrogate end points in clinical trials supporting approval of drugs for cancer treatment by the Japanese regulatory agency - Annals of Oncology

Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics

COVID‐19 drug repurposing: A review of computational screening methods, clinical trials, and protein interaction assays - Wang - 2021 - Medicinal Research Reviews - Wiley Online Library

The changing landscape of anti-lymphoma drug clinical trials in mainland China in the past 15 years (2005–2020): A systematic review - The Lancet Regional Health – Western Pacific
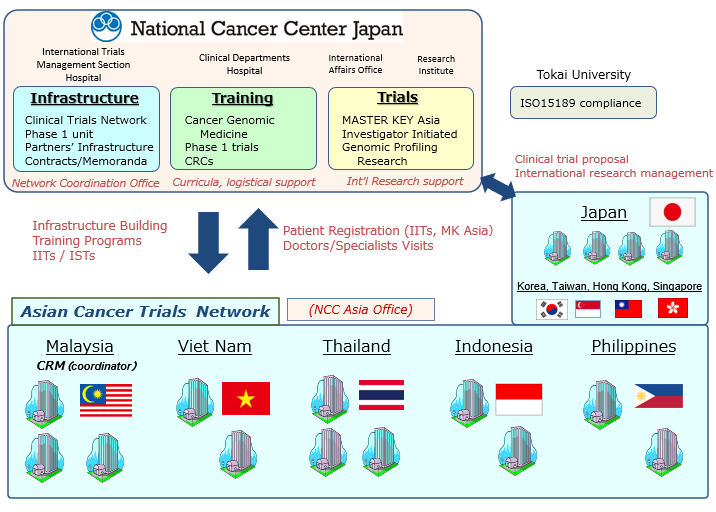
ATLAS Project – Paving the way towards developing drugs with Asian partners | National Cancer Center Japan

Changes in clinical trials of cancer drugs in mainland China over the decade 2009–18: a systematic review - The Lancet Oncology






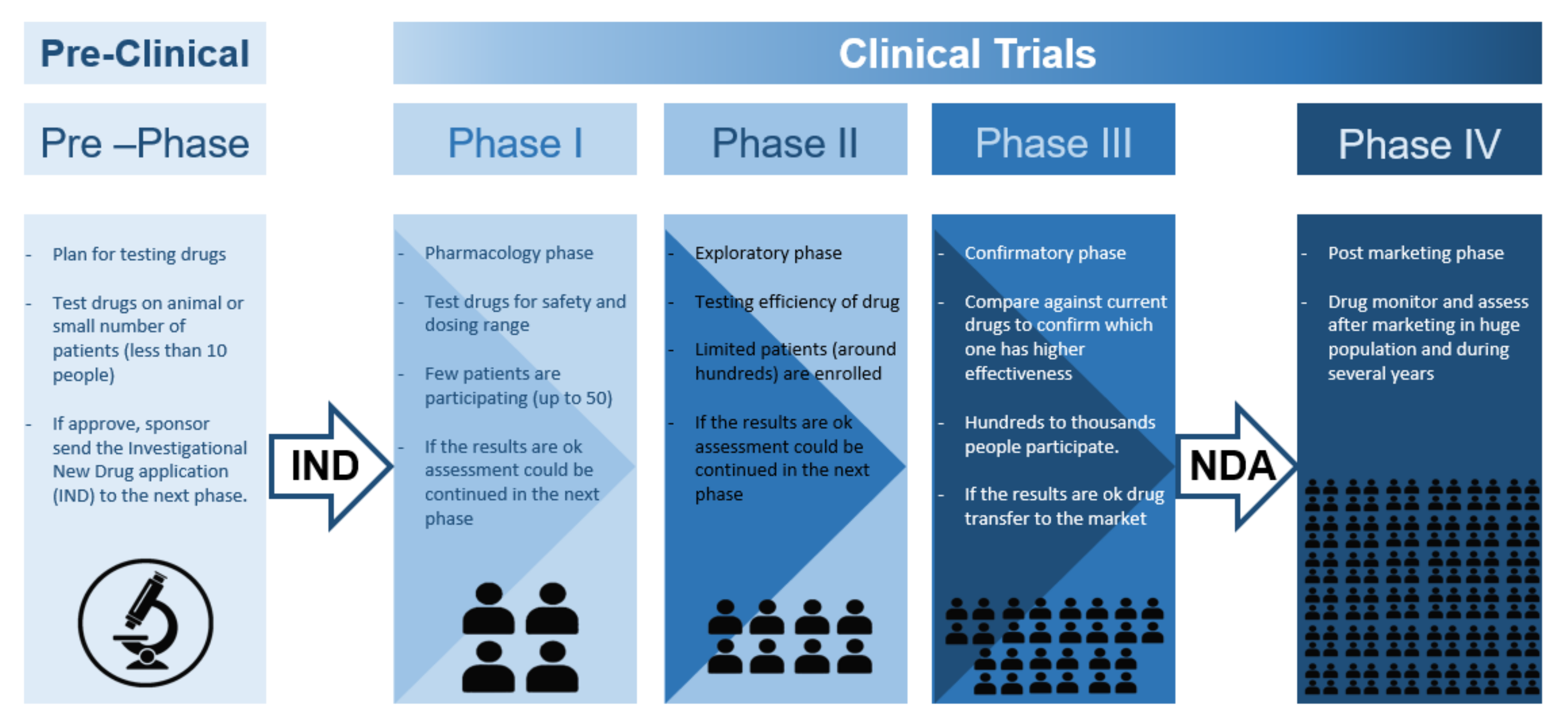

![Stages of PWS Drug Development Overview [VIDEO] Stages of PWS Drug Development Overview [VIDEO]](https://www.fpwr.org/hs-fs/hubfs/Clinical%20Trials/Drug%20Development%20Process/Slide5.jpeg?width=459&name=Slide5.jpeg)