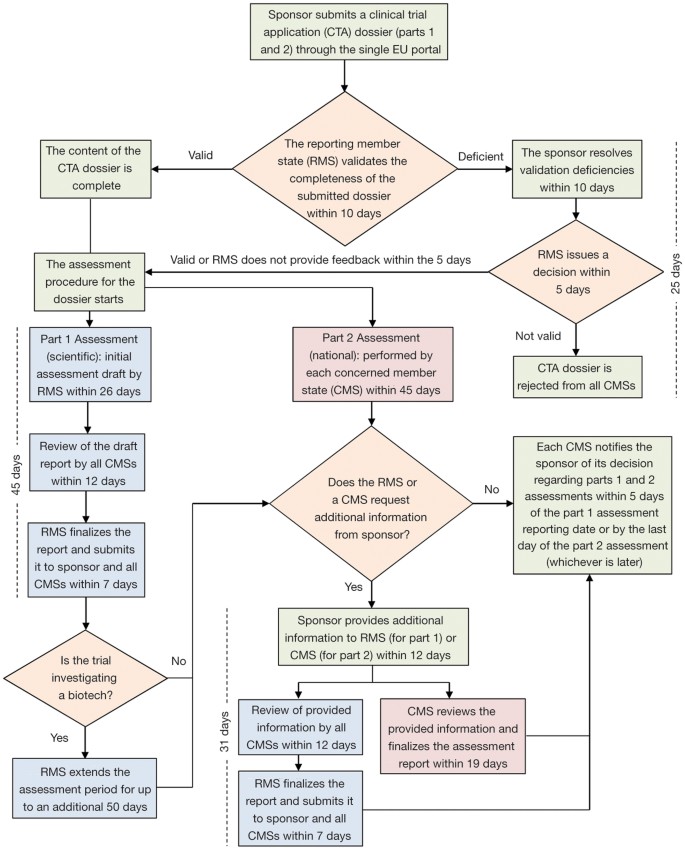
When innovation outpaces regulations: The legal challenges for direct‐to‐patient supply of investigational medicinal products - Malone - 2022 - British Journal of Clinical Pharmacology - Wiley Online Library

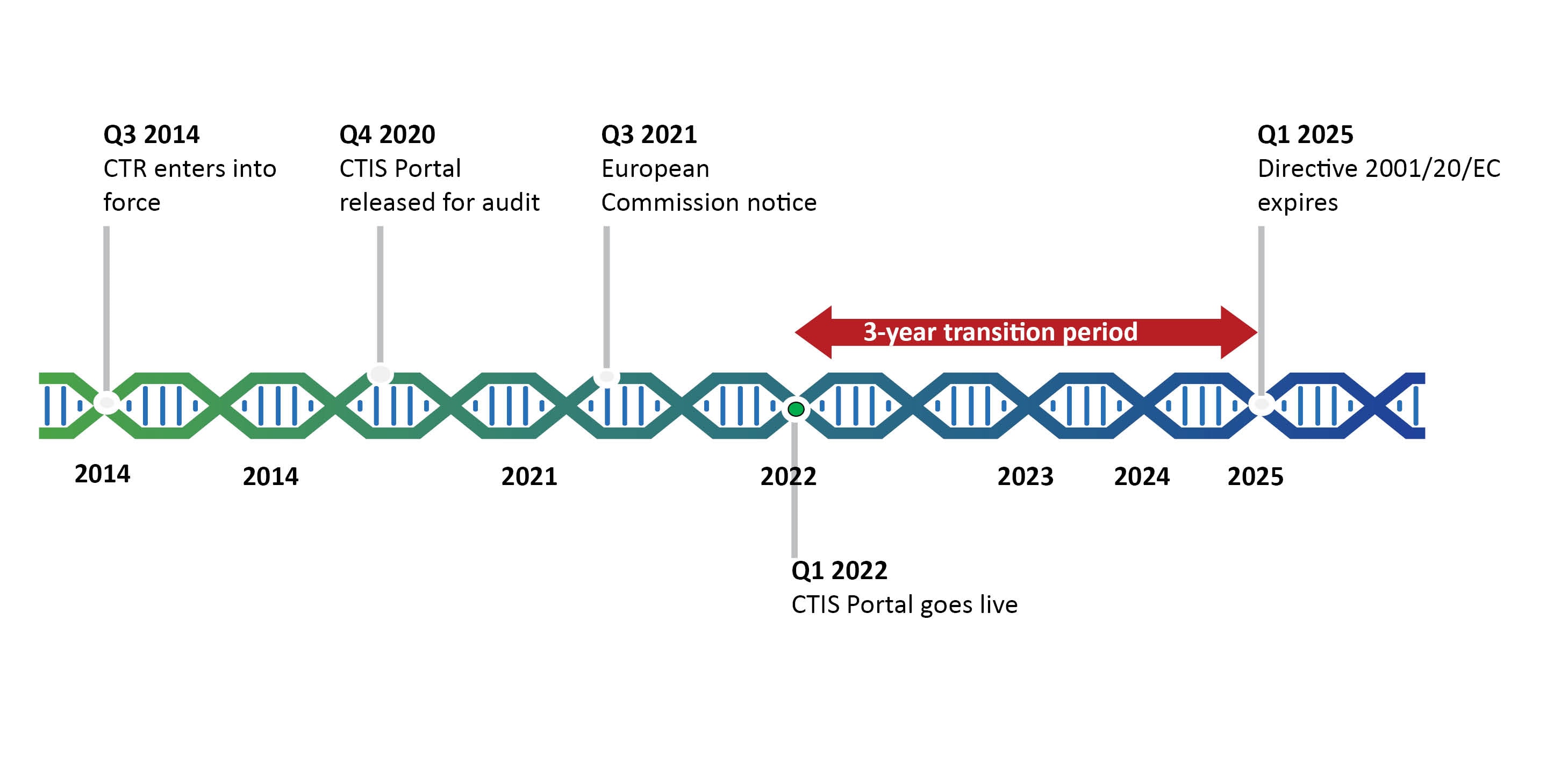
EU Clinical Trial Regulation 536/2014potential timeline. EoT end of... | Download Scientific Diagram
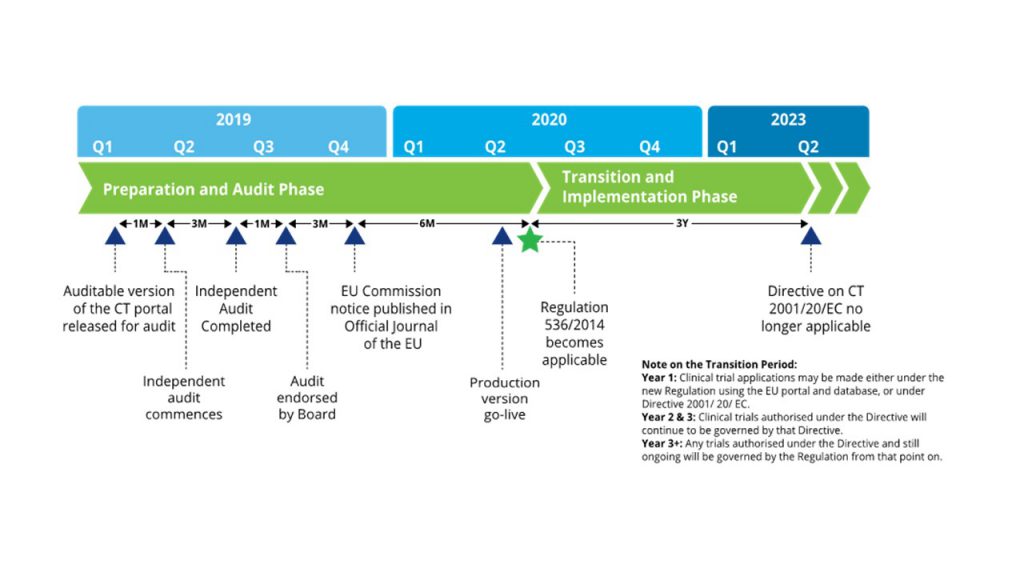
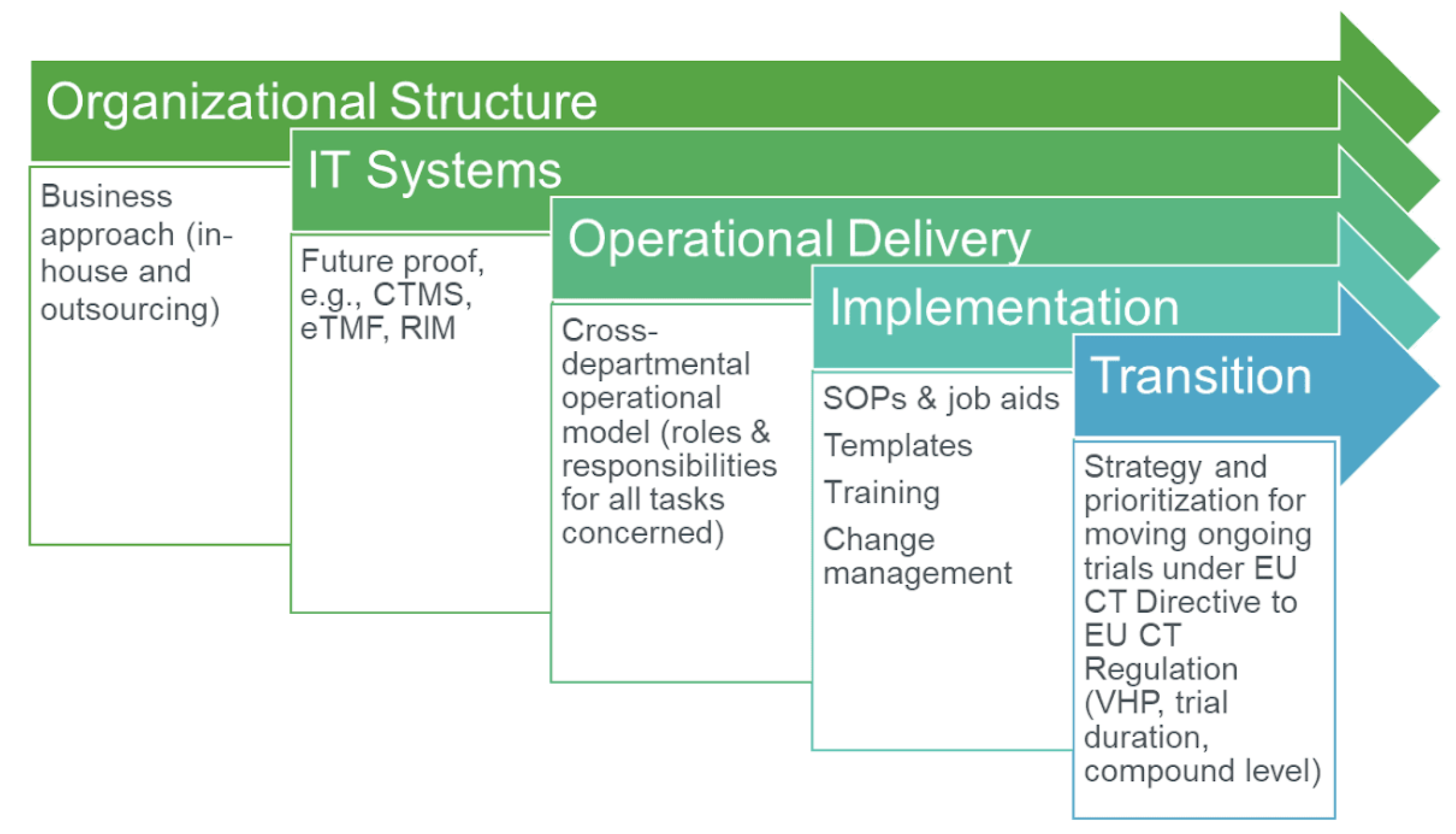
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS

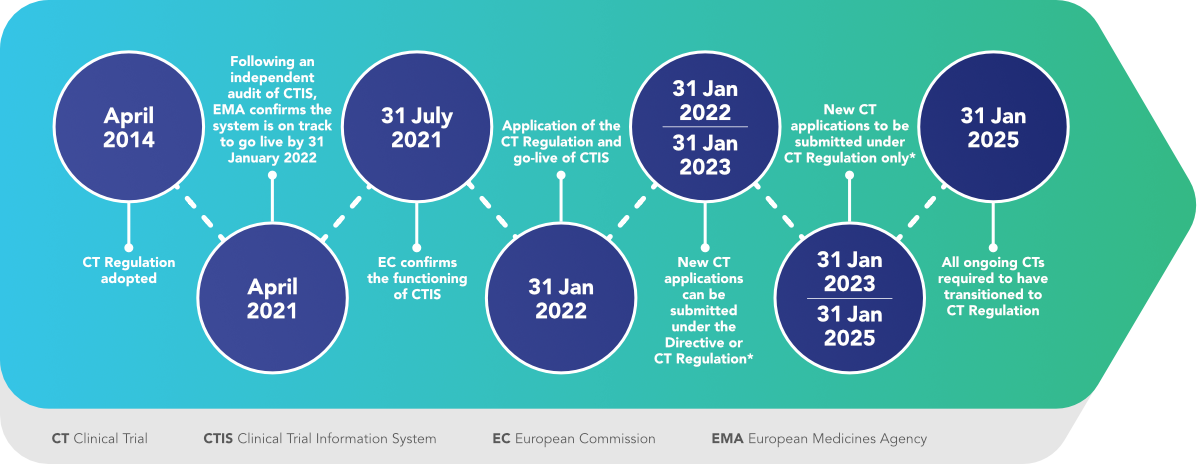
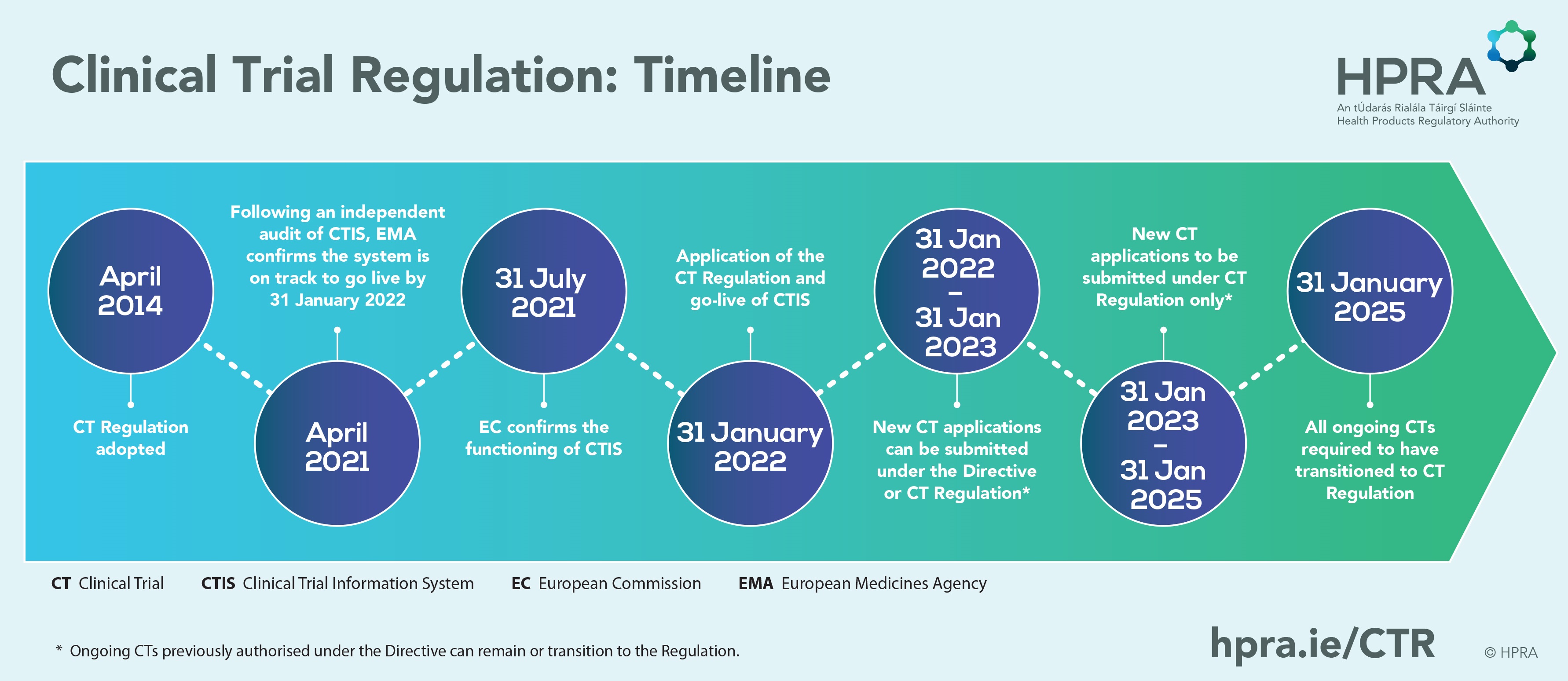
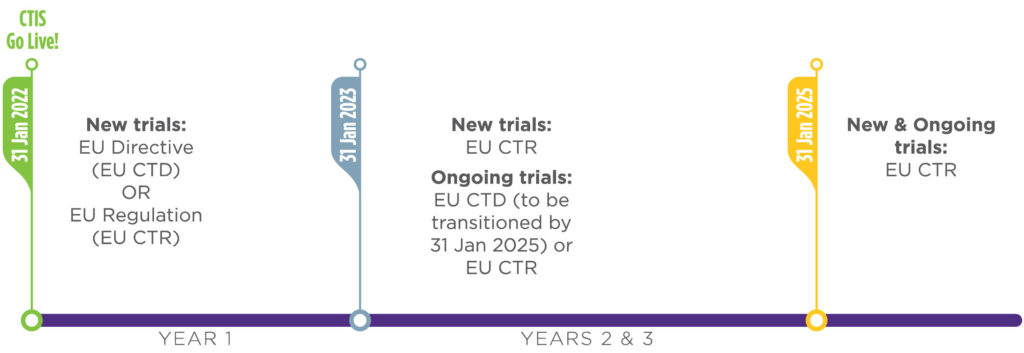
Managing the New EU Clinical Trials Regulation 536/2014 – Guidance for Navigating the Clinical Trial Information System (CTIS)