Fixed-Dose Combinations Improve Medication Compliance: A Meta-Analysis - The American Journal of Medicine
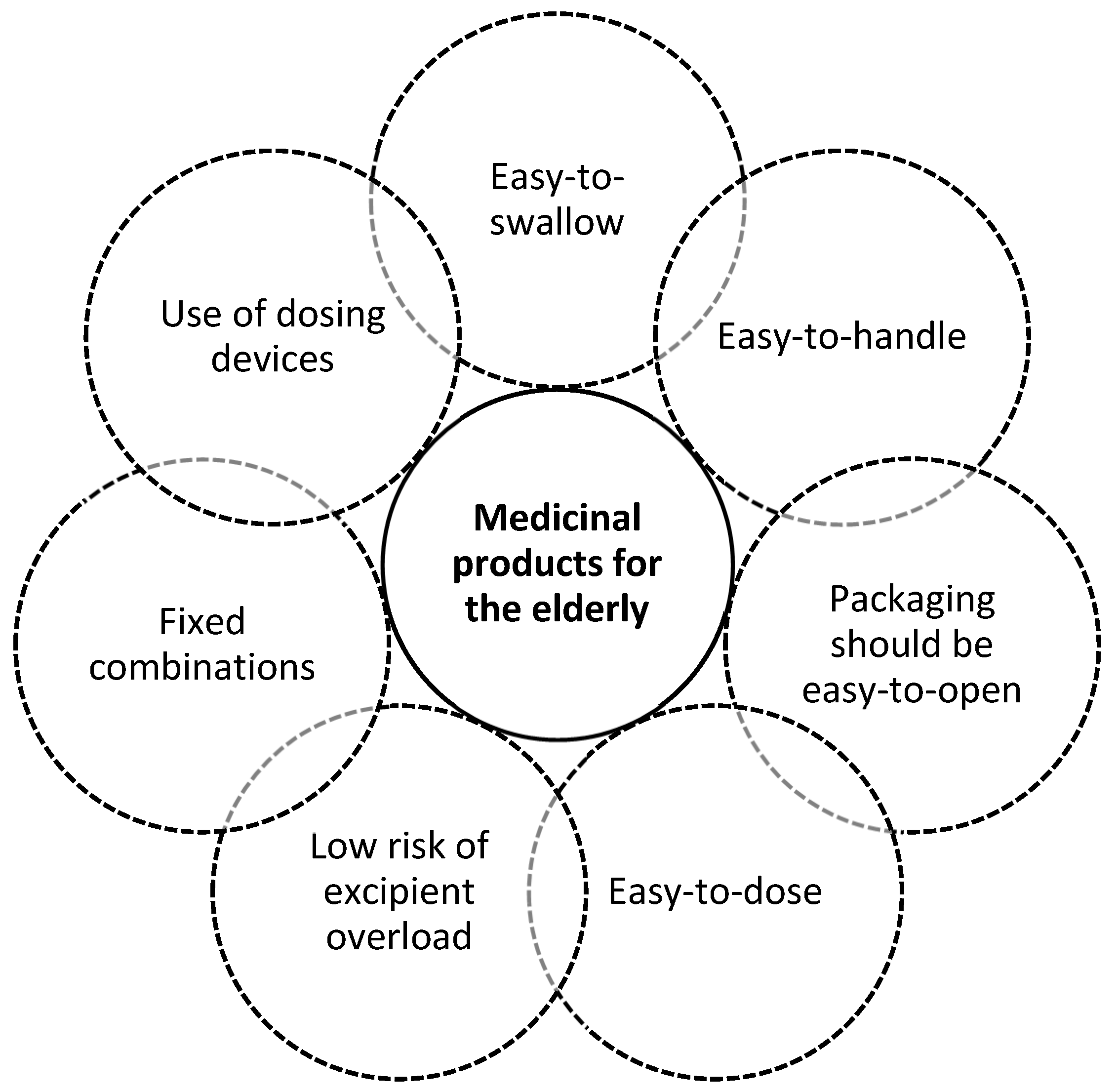
Pharmaceutics | Free Full-Text | Patient Centric Pharmaceutical Drug Product Design—The Impact on Medication Adherence | HTML

New Draft EMA-Guideline On Clinical Development Of Fixed Combination Medicines | The National Law Review

Annex 5 Guidelines for registration of fixed-dose combination medicinal products - PDF Free Download

Initial treatment with a single pill containing quadruple combination of quarter doses of blood pressure medicines versus standard dose monotherapy in patients with hypertension (QUARTET): a phase 3, randomised, double-blind, active-controlled trial -

Pharmaceutical application and development of fixed-dose combination: dosage form review | SpringerLink
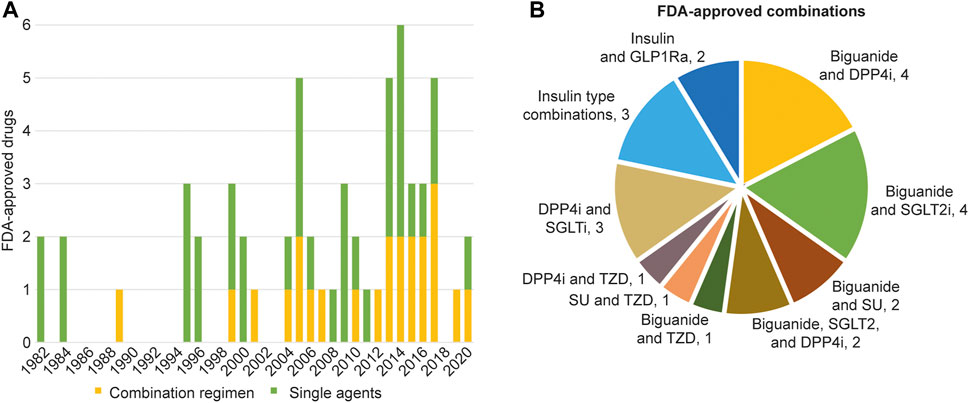
Frontiers | Trends in Antidiabetic Drug Discovery: FDA Approved Drugs, New Drugs in Clinical Trials and Global Sales

Fixed-dose combination therapies with and without aspirin for primary prevention of cardiovascular disease: an individual participant data meta-analysis - The Lancet

Regulatory guidelines and preclinical tools to study the biodistribution of RNA therapeutics - ScienceDirect

Challenges and opportunities to include patient‐centric product design in industrial medicines development to improve therapeutic goals - Timpe - 2020 - British Journal of Clinical Pharmacology - Wiley Online Library