Frontiers | A Companion Diagnostic With Significant Clinical Impact in Treatment of Breast and Gastric Cancer | Oncology

Clinical outcomes of patients with breast cancer relapsing after (neo)adjuvant trastuzumab and receiving trastuzumab rechallenge or lapatinib-based therapy: a multicentre retrospective cohort study - ESMO Open

Subcutaneous versus intravenous administration of (neo)adjuvant trastuzumab in patients with HER2-positive, clinical stage I–III breast cancer (HannaH study): a phase 3, open-label, multicentre, randomised trial - The Lancet Oncology

Schema of the APHINITY clinical trial evaluating efficacy of adjuvant... | Download Scientific Diagram

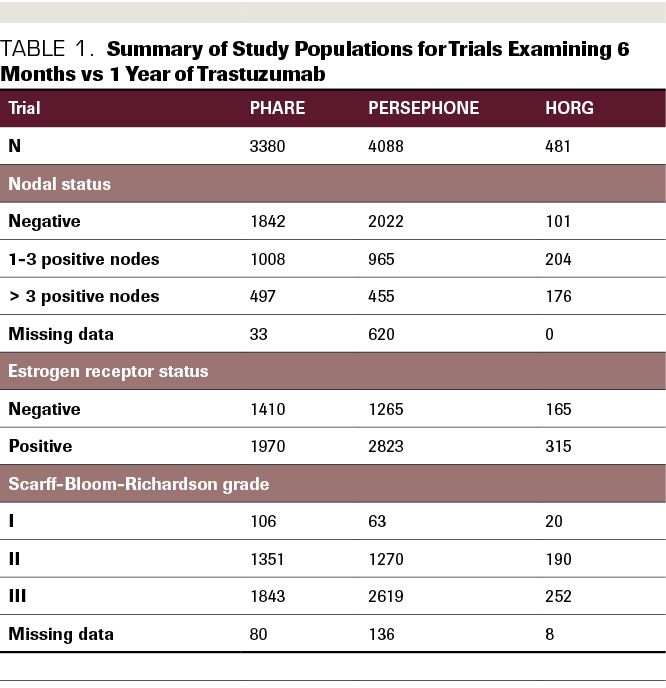
6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (PERSEPHONE): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial - The Lancet

Final Efficacy Results of Neratinib in HER2-positive Hormone Receptor-positive Early-stage Breast Cancer From the Phase III ExteNET Trial - Clinical Breast Cancer
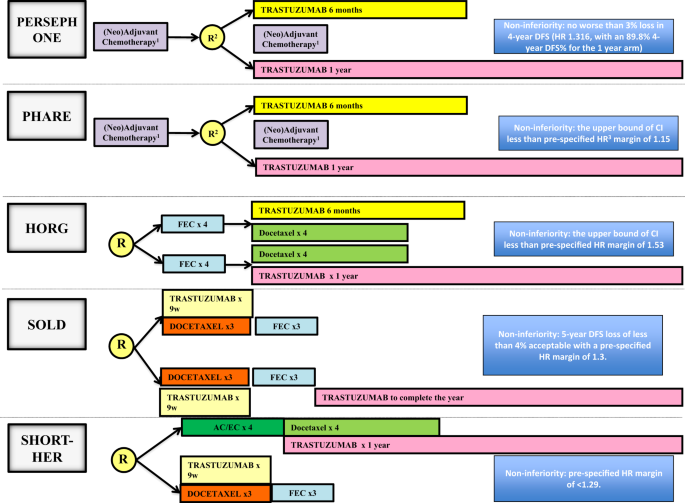
PERSEPHONE: are we ready to de-escalate adjuvant trastuzumab for HER2-positive breast cancer? | npj Breast Cancer
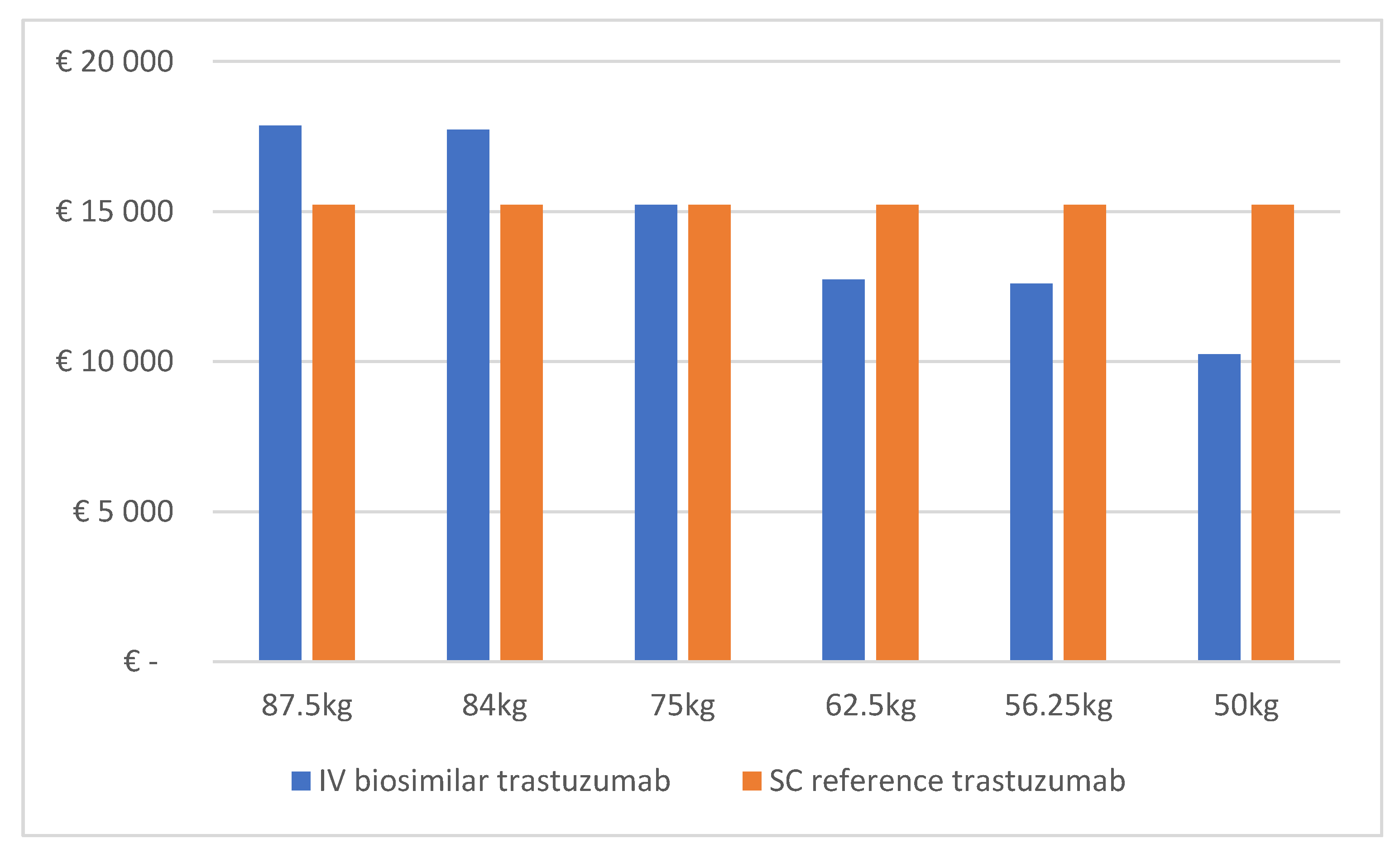
Pharmaceuticals | Free Full-Text | Simulating Costs of Intravenous Biosimilar Trastuzumab vs. Subcutaneous Reference Trastuzumab in Adjuvant HER2-Positive Breast Cancer: A Belgian Case Study | HTML

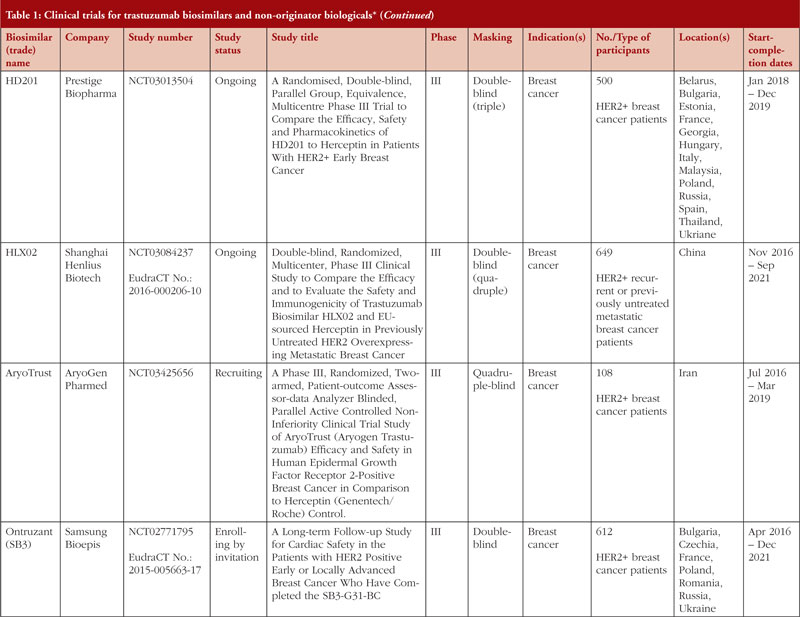
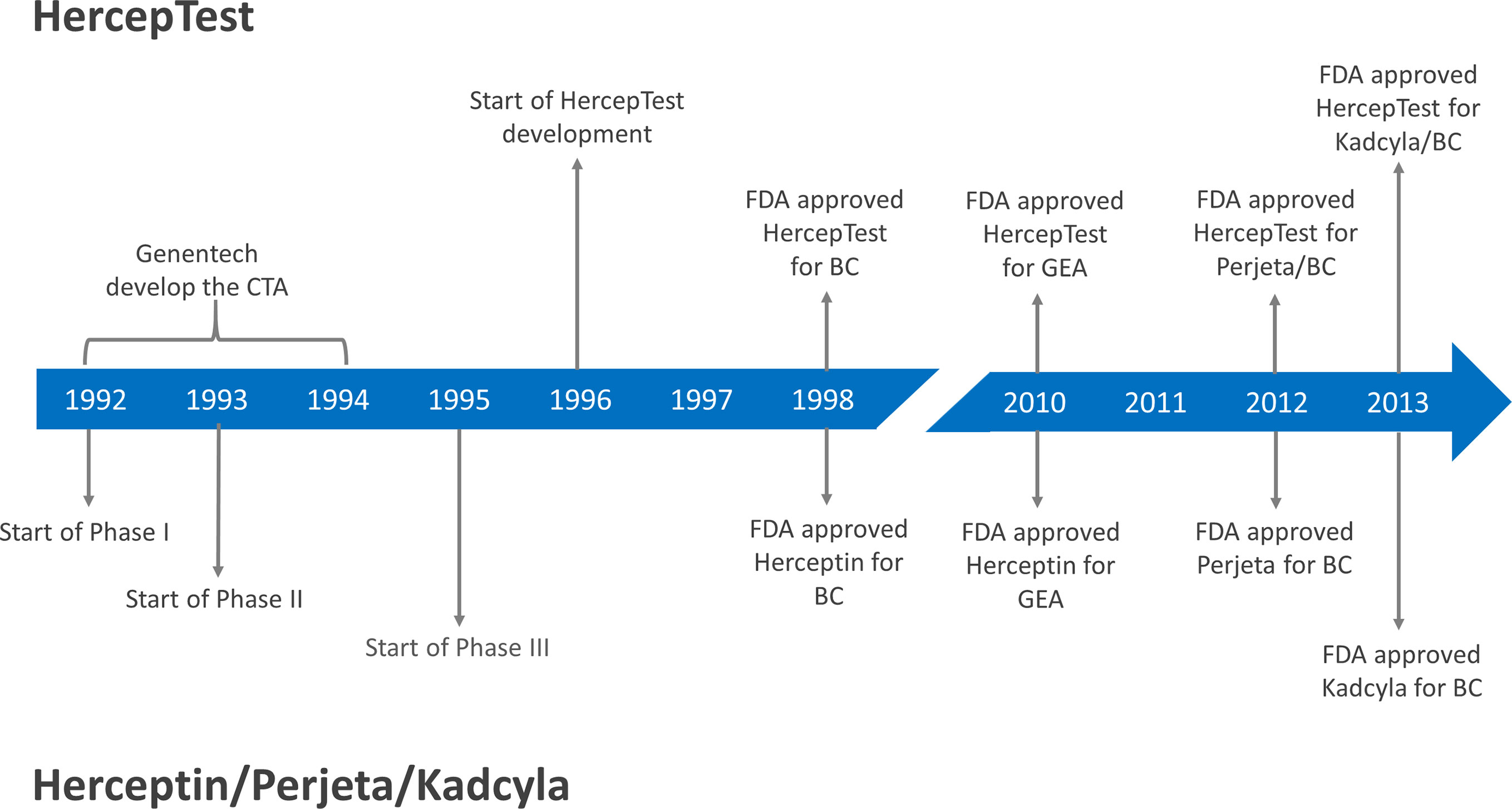
![PDF] Clinical trials of Herceptin(trastuzumab). | Semantic Scholar PDF] Clinical trials of Herceptin(trastuzumab). | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4d6467fad15262096d78d0838ef39414b4b4d90a/2-Table1-1.png)


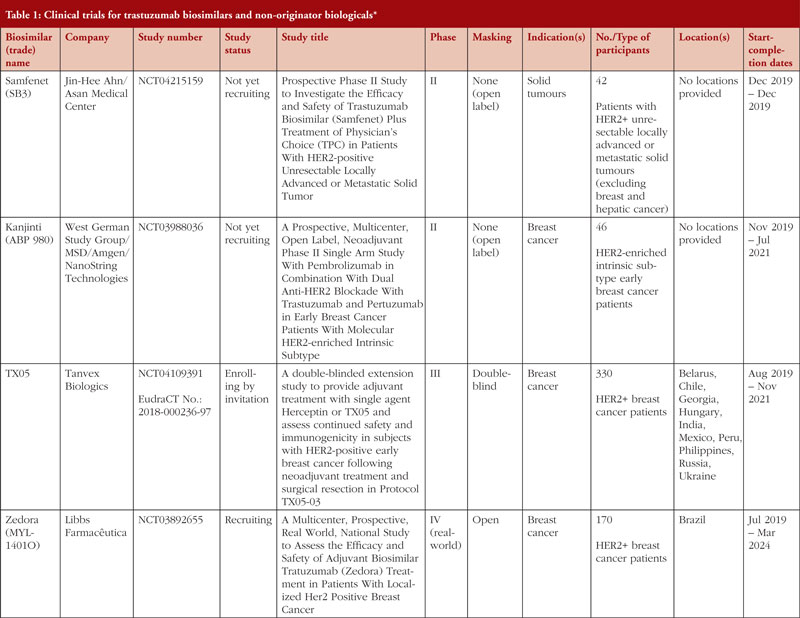

/herceptin-biologic-therapy-for-breast-cancer-430573-FINAL2-ace372d34fd544149e92f294c2c22958.png)