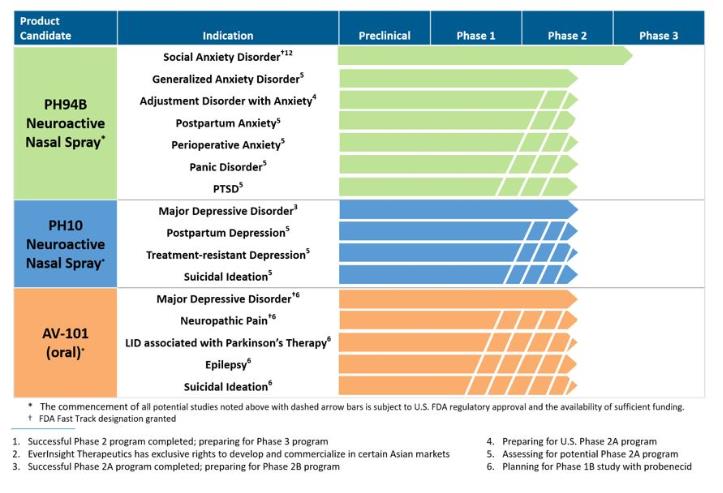
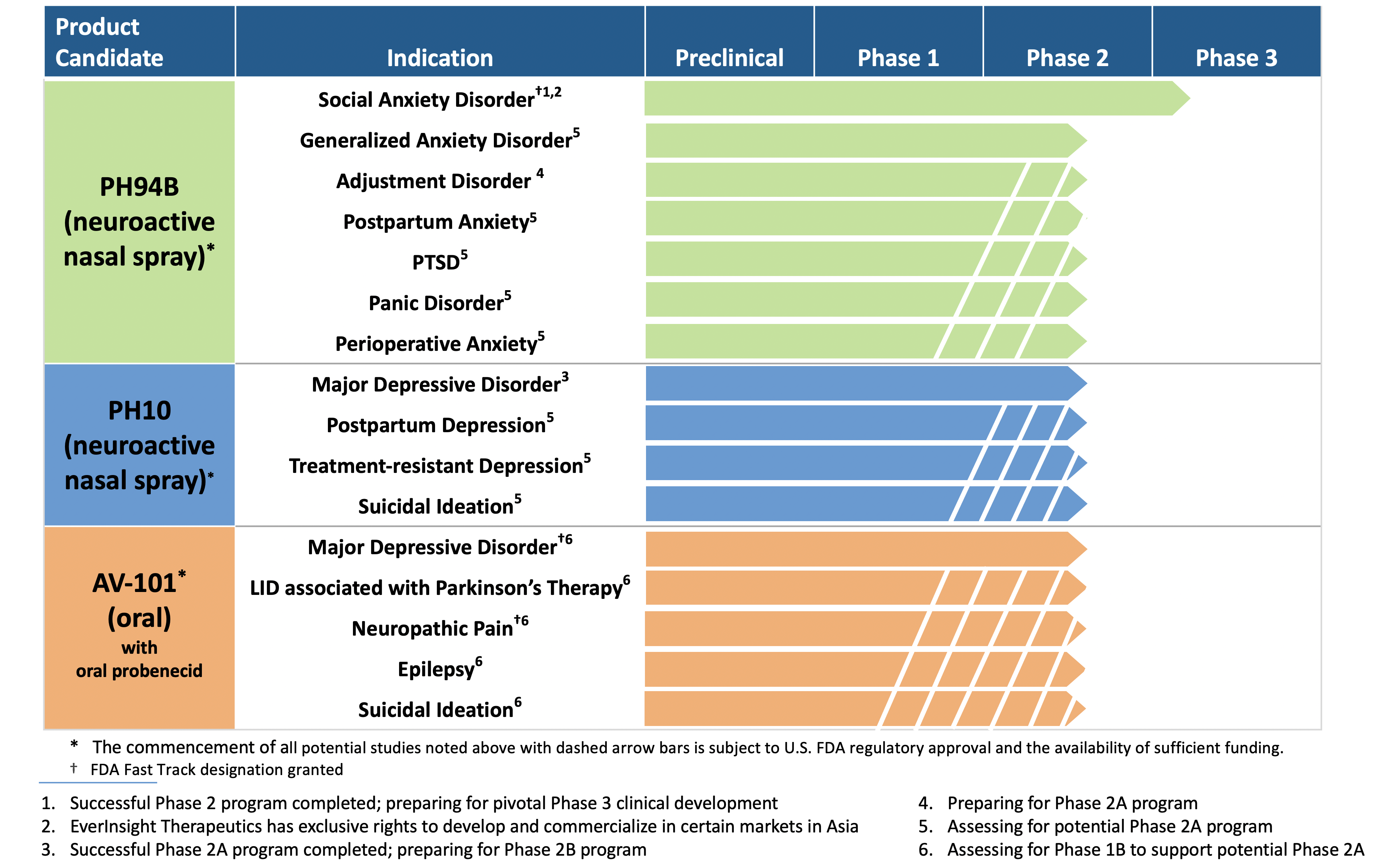
VistaGen to Present PH94B Exploratory Phase 2A Research Program for Adjustment Disorder with Anxiety at American Society for Clinical Psychopharmacology Annual Meeting

PDF) Effect of as-needed use of intranasal PH94B on social and performance anxiety in individuals with social anxiety disorder: L iebowitz et al.

VistaGen Reports Positive Preclinical Data Differentiating Mechanism of Action of PH94B from Risk-Ridden Benzodiazepines

VistaGen readies Phase 3 trial of novel, fast-acting neuroactive nasal spray in social anxiety disorder - BioTuesdays

VistaGen and AffaMed Complete Key Regulatory Submissions for PALISADE Global Phase 3 Clinical Trial for PH94B | Business Wire

PH94B Neuroactive Nasal Spray, a Potential On-Demand Treatment for Social Anxiety Disorder | Learn how PH94B, a novel, rapid-acting CNS neuroactive nasal spray being developed by VistaGen Therapeutics for the treatment of

VistaGen says FDA found no signal of abuse potential for its nasal spray PH94B for anxiety | Seeking Alpha


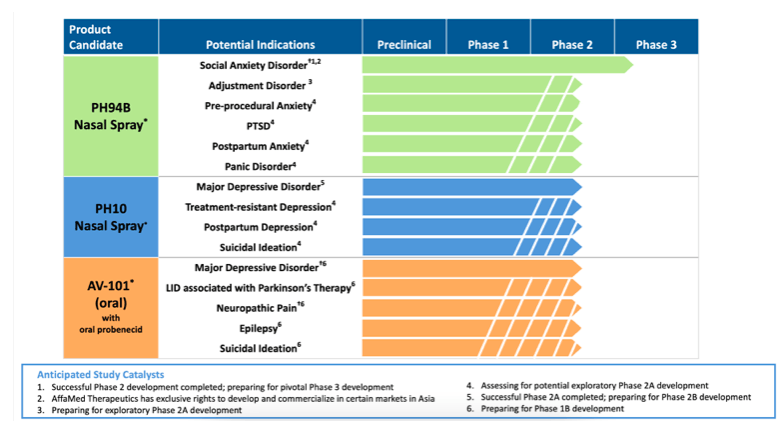

,%20Phobia,%20Social,%20Anxiety%20Disorders:%0A%0APH94B%20Nasal%20Spray%20for%20Social%20Anxiety%20Disorder%20(SAD).png?md=1)