IJMS | Free Full-Text | Neurotoxic Soluble Amyloid Oligomers Drive Alzheimer's Pathogenesis and Represent a Clinically Validated Target for Slowing Disease Progression | HTML
Trial registration, publication rate and characteristics in the research field of otology: A cross-sectional study | PLOS ONE

Publication and reporting of clinical trial results: cross sectional analysis across academic medical centers | The BMJ

Requirements for Registering & Reporting NIH-funded Clinical Trials in ClinicalTrials.gov | grants.nih.gov

Acer Therapeutics Enrolls First Patient in its Phase 2a Clinical Trial of ACER-801 for Treatment of Moderate to Severe Vasomotor Symptoms Associated with Menopause

FOCUS ON NLM RESOURCES: CLINICALTRIALS.GOV. WHAT IS A CLINICAL STUDY? Research study using human subjects. Volunteers may have a certain disease or. - ppt download

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH
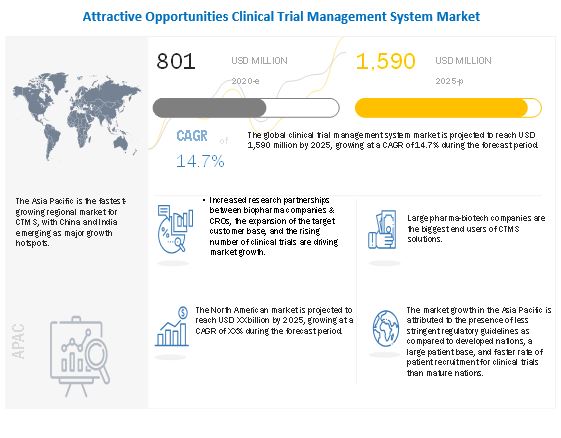
Clinical Trial Management System Market | (2022 - 2025) | Size, Share and Trends | MarketsandMarkets