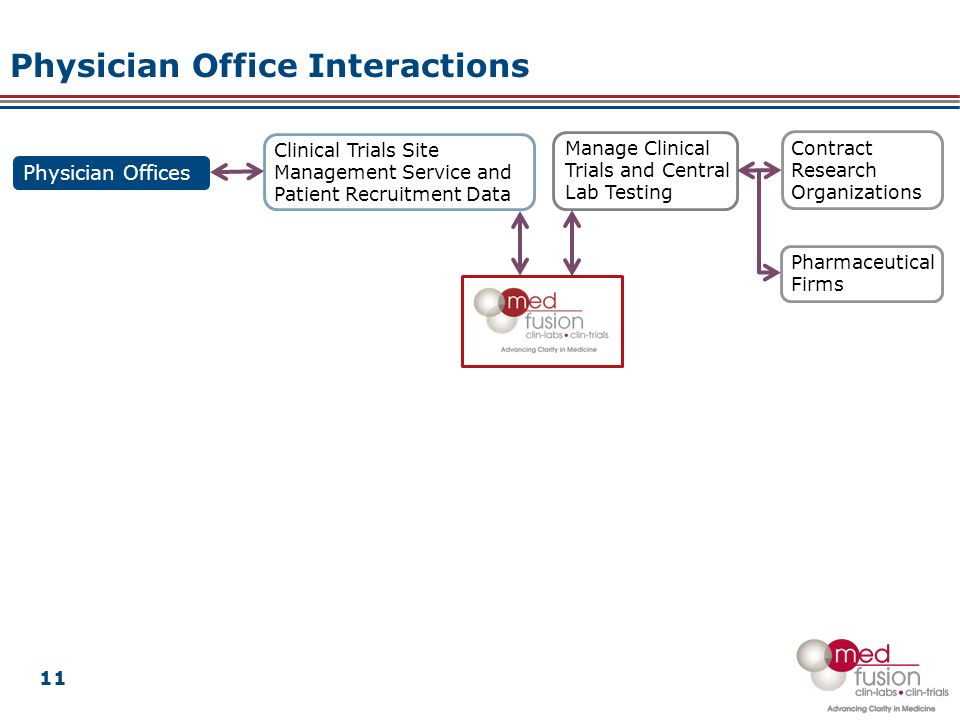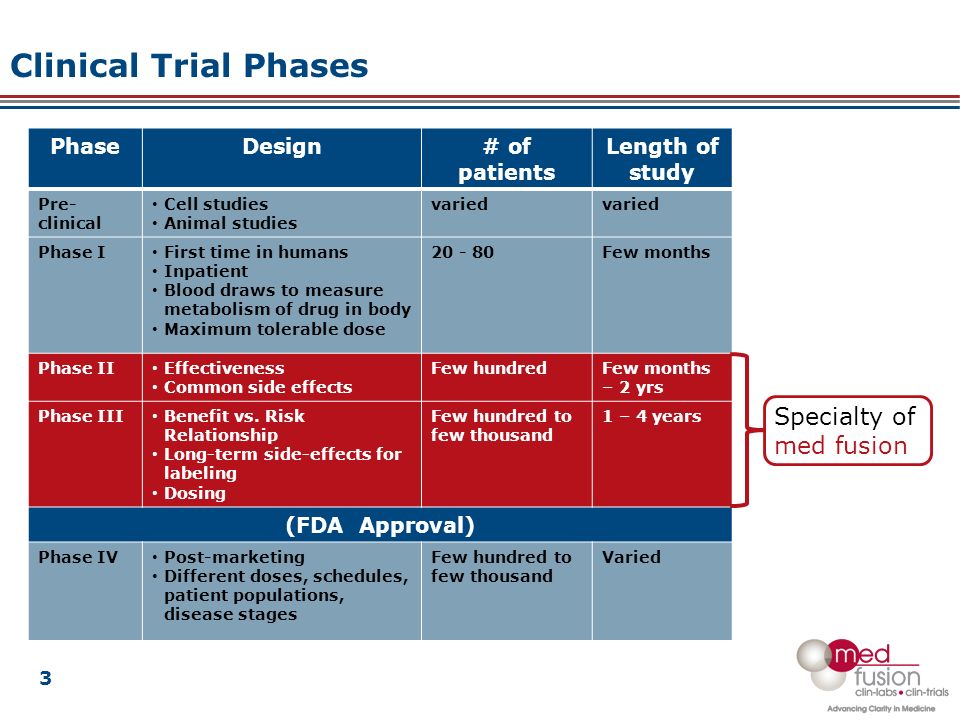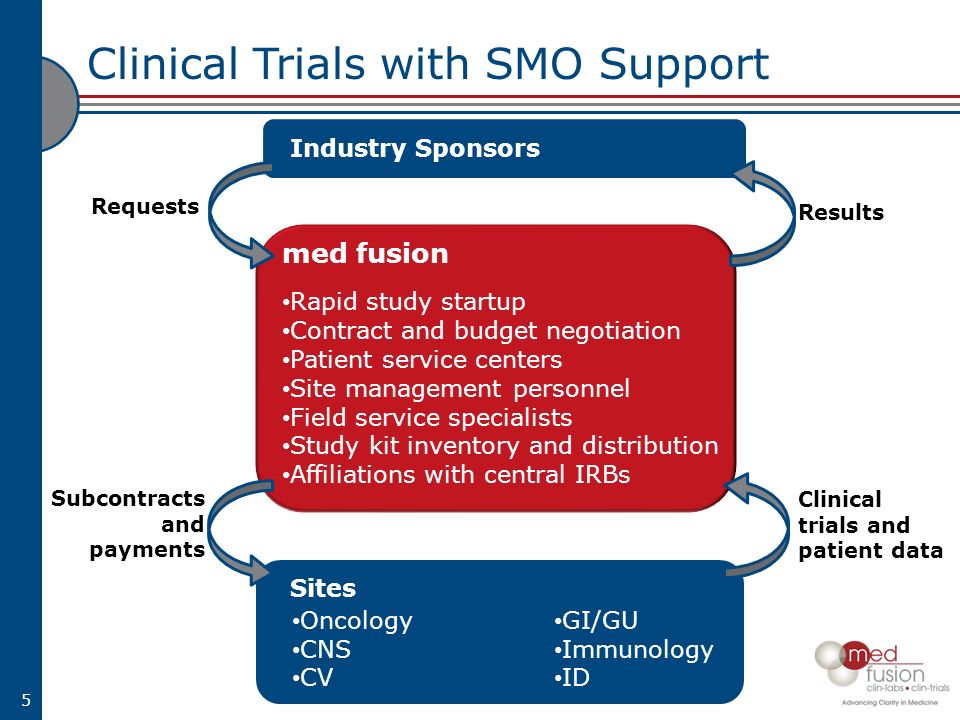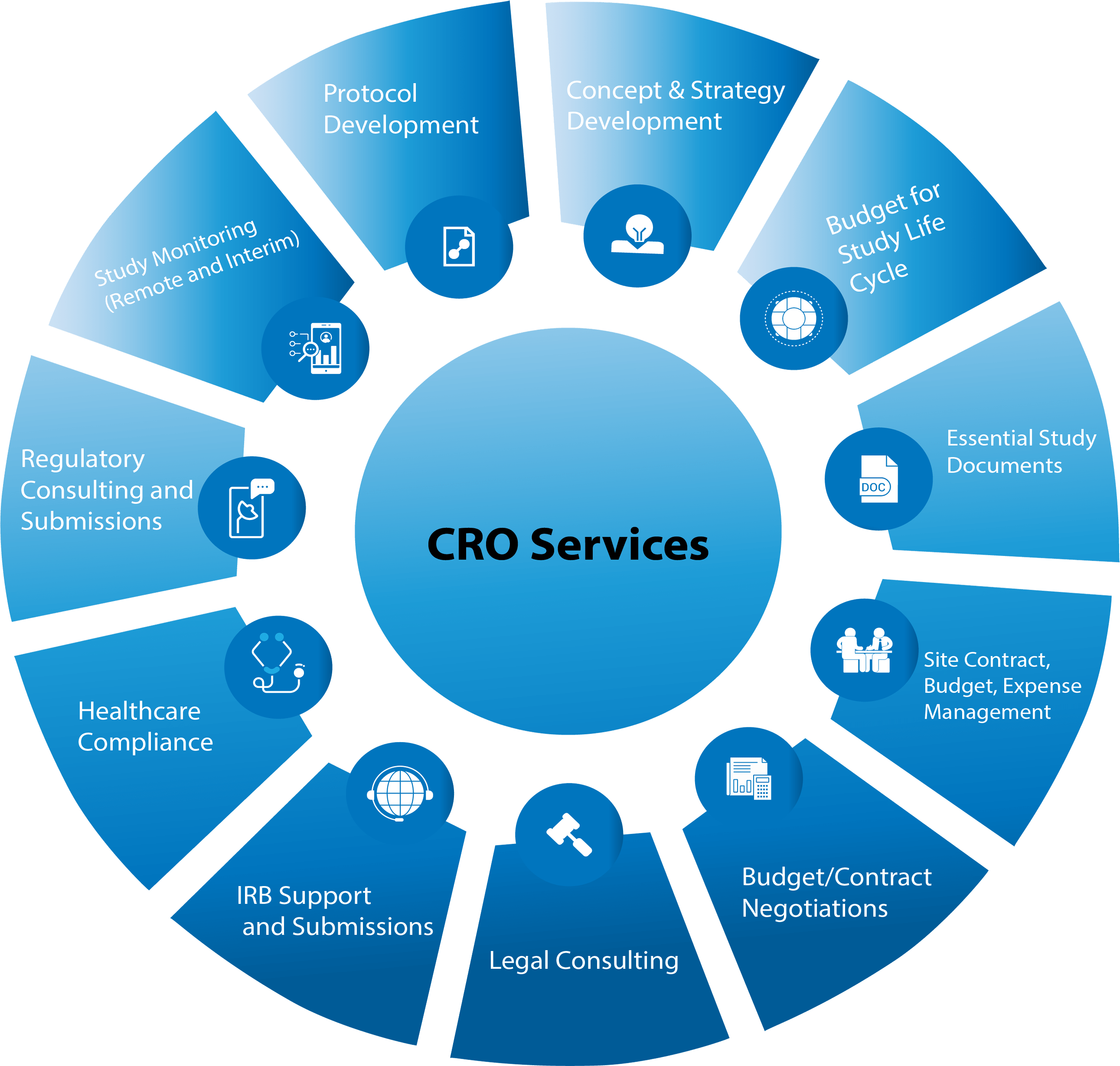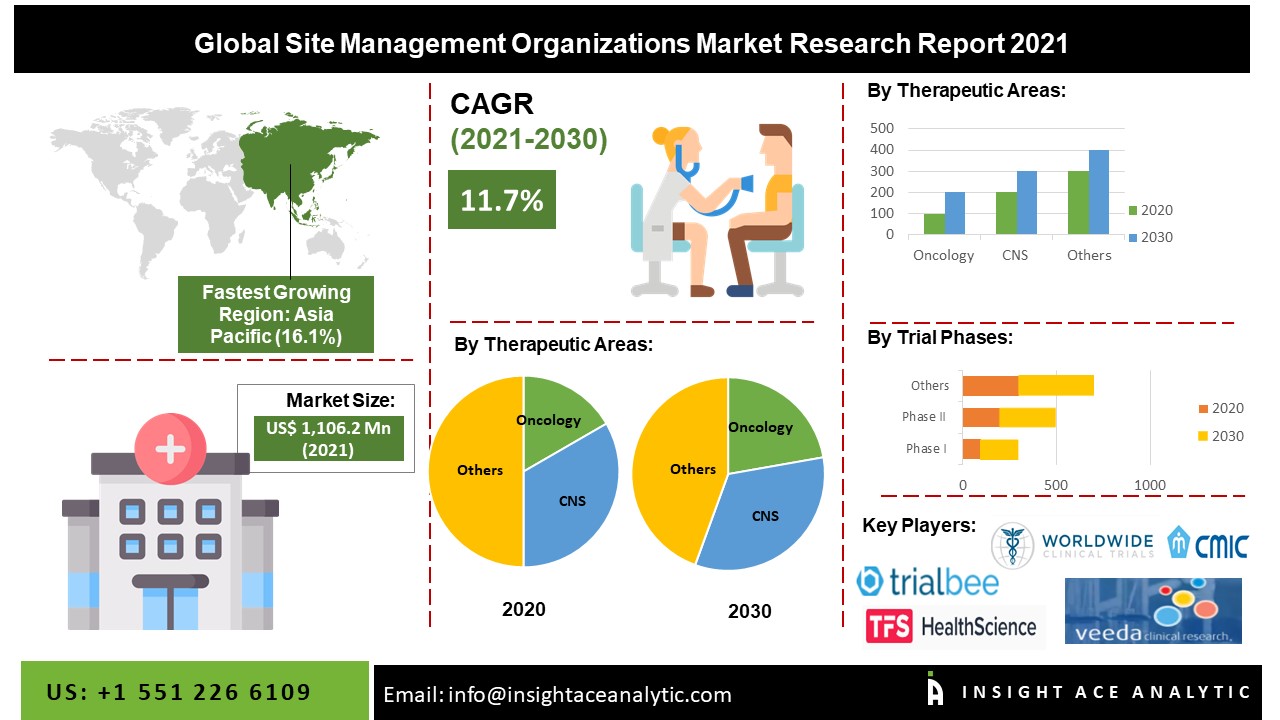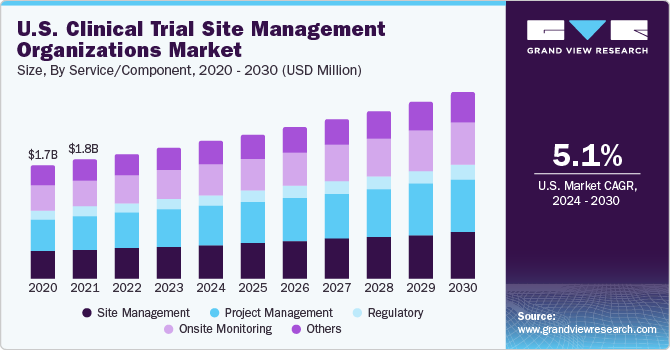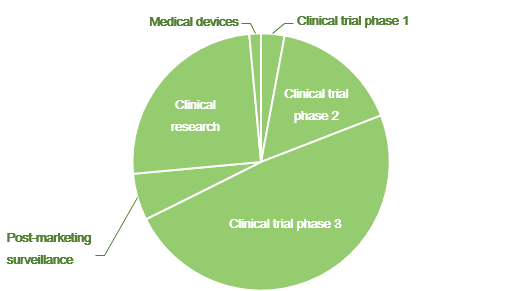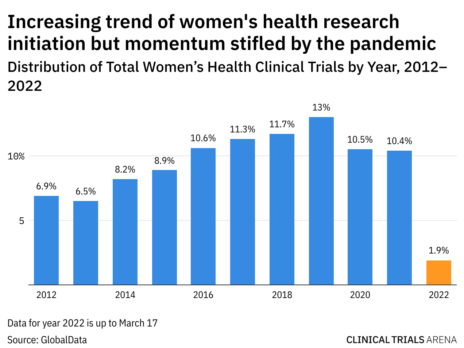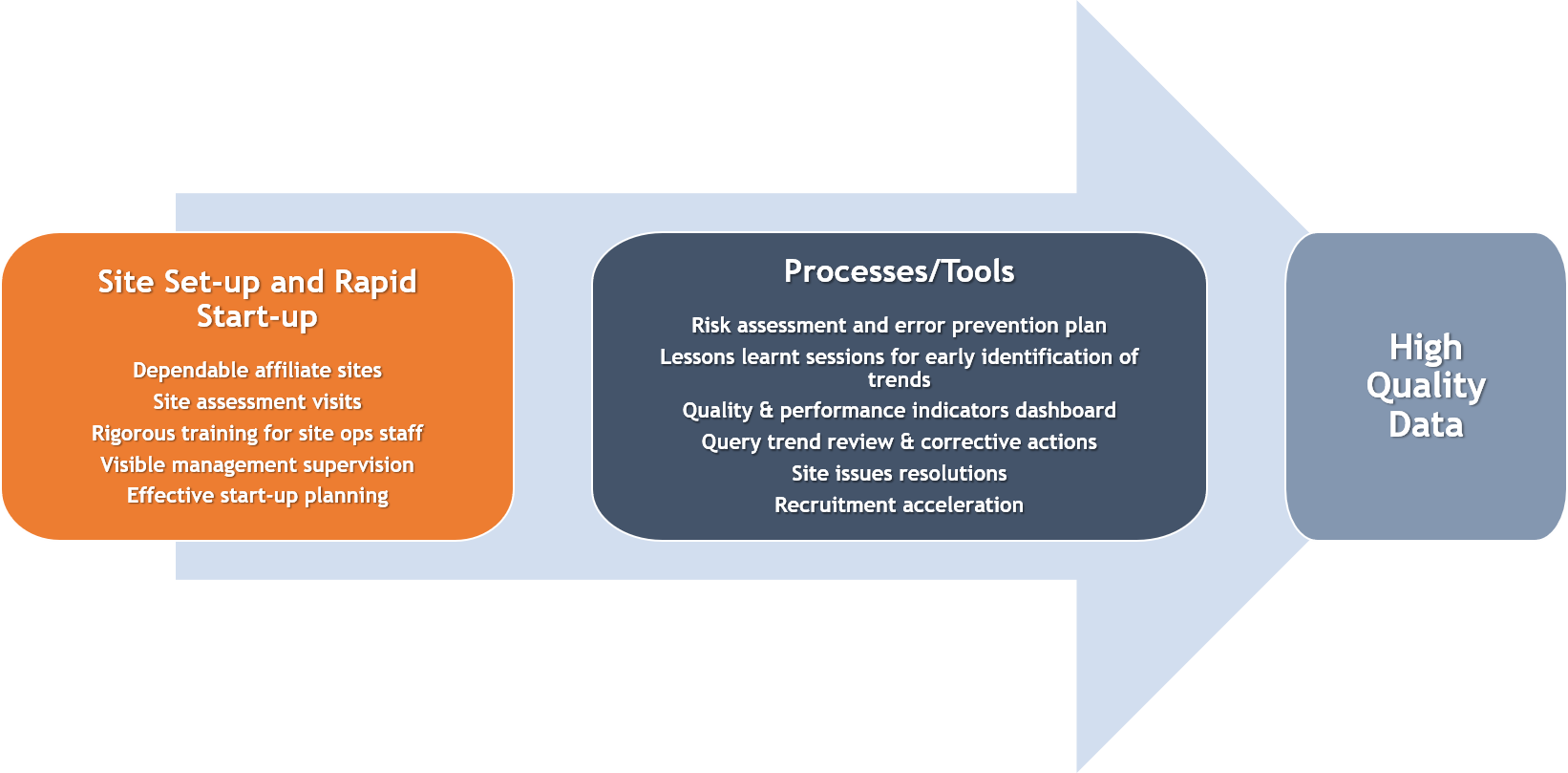
Overall Study Management | S4-Research – Site Support Services for Clinical Trials I Site Management Organization I SMO India I Site Management Organisation I SMO Asia I SMO India I SMO I

Considerations for Third Party Vendor Management in a Risk-Focused Environment | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

CLINOVEX Clinical Research Solution - REGISTRATION OPEN CLINOVEX is pleased to announce the Admission Notification of 15th Batch “Diploma in Clinical Research management (DCRM)” with 100% placement. Our candidates are employed in
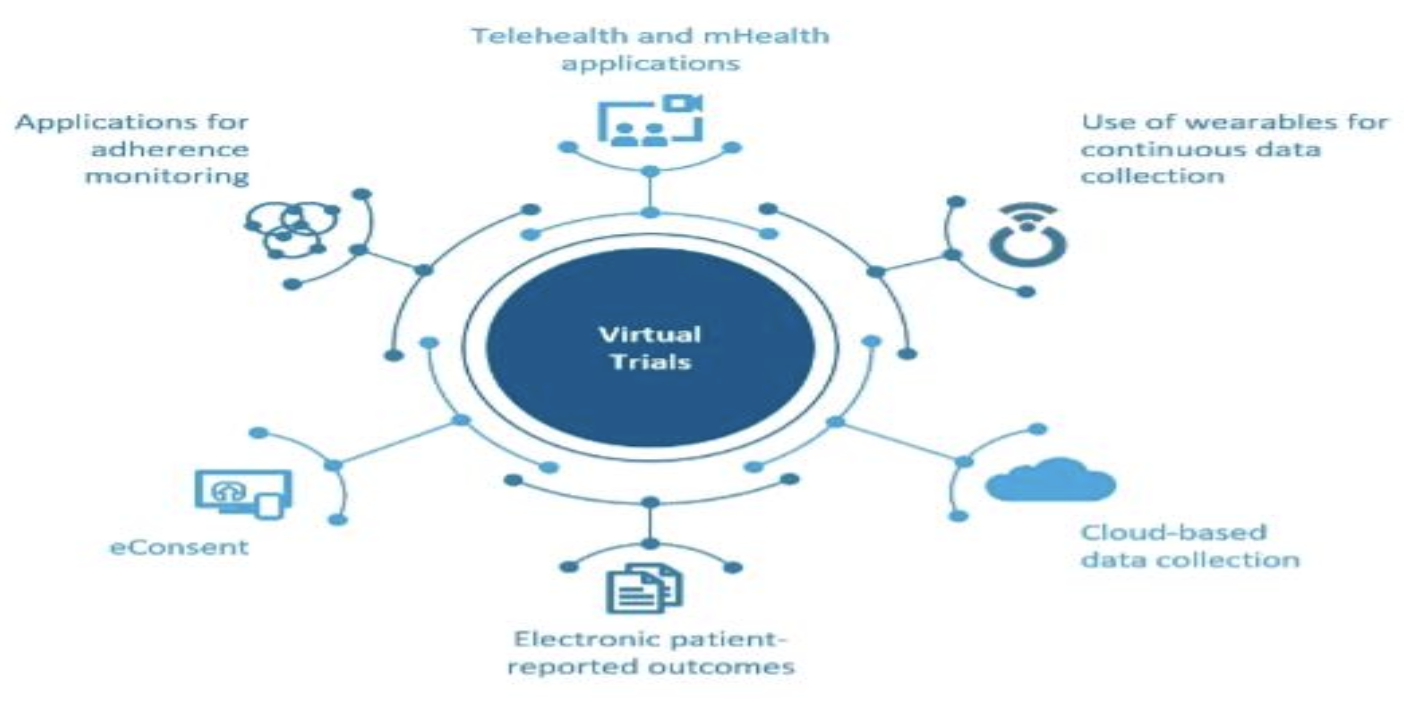
Career — Clinical Research Blog | Certified Clinical Research Professionals Society - Clinical Research Certification